|
| Topics on Continuous Training |
J. Martín Ruano*![]() , L. Alcubilla García**
, L. Alcubilla García**
*Pediatrics. San Juan Health Center. Salamanca
**Pediatrics. Salamanca University Hospital Complex
| Abstract
Acute bronchiolitis (AB) is the first episode of respiratory distress in an infant under 24 months of age. It is the most common acute lower respiratory tract infection in children under one year old. Respiratory syncytial virus (RSV) infection in children almost always produces clinical symptoms, which are generally benign and self-limiting, with variable expression depending on the patient’s age, risk factors, or previous exposure to the virus. It is a seasonal and epidemic disease, with a higher incidence in the colder months. The most frequent cause of AB is viral infection, with the responsible viruses varying depending on the season and geographic area. RSV is the most frequently detected virus, responsible for approximately 70% of cases. The second most frequent is rhinovirus (RV). The disease begins with upper respiratory tract symptoms lasting one to three days. The infection then spreads to the lower respiratory tract, where symptoms become more pronounced, including cough and signs of respiratory distress such as tachypnea, wheezing, and crackles. Diagnosis is clinical, and further testing is usually unnecessary. Treatment is primarily supportive; Adequate hydration and oxygenation remain the cornerstone of clinical management. Current clinical practice guidelines advise against the routine use of medications such as bronchodilators, corticosteroids, or antibiotics. Thanks to immunization with nirsevimab, administered to over 90% of infants under six months of age in 2023, hospital admissions for acute bronchiolitis were reduced by up to 80%. Pediatric bronchitis includes acute episodes, predominantly viral, and persistent forms associated with bacterial infection, notably protracted bacterial bronchitis (PBB). Acute bronchitis is primarily characterized by cough and may be accompanied by mild constitutional symptoms with a self-limited course; diagnosis is clinical and management is symptomatic with supportive care. In contrast, PBB is defined by a chronic productive cough lasting more than 3-4 weeks, with an appropriate response to a prolonged course of broad-spectrum antibiotics. Lack of therapeutic response warrants consideration of alternative diagnoses such as bronchiectasis. |
| Resumen
La bronquiolitis aguda (BA) es el primer episodio de dificultad respiratoria en un lactante menor de 24 meses. Es la infección aguda de vías respiratorias bajas más frecuente en niños menores de un año. La infección por virus respiratorio sincitial (VRS) en los niños casi siempre va a producir sintomatología clínica, siendo generalmente benigna y autolimitada y variando su expresividad según la edad del paciente, factores de riesgo o exposiciones previas al virus. Es una enfermedad estacional y epidémica, con mayor incidencia en los meses fríos. La causa más frecuente de la BA son las infecciones víricas, variando los virus responsables en función de la estación del año y la zona geográfica. El VRS es el virus detectado con mayor frecuencia, siendo responsable de aproximadamente el 70 % de los casos. El segundo en frecuencia es el rhinovirus (RV). La enfermedad comienza con síntomas respiratorios de vías altas durante uno a tres días. A continuación, la infección desciende a vías respiratorias bajas, donde los síntomas se vuelven más evidentes con tos y signos de dificultad respiratoria, como taquipnea, sibilancias y crepitantes. El diagnóstico es clínico y, habitualmente, no será necesaria la realización de pruebas complementarias. El tratamiento es fundamentalmente de soporte; una buena hidratación y una adecuada oxigenación siguen siendo la base del manejo clínico. Las guías de práctica clínica actuales desaconsejan el uso rutinario de medicamentos, como broncodilatadores, corticoides o antibióticos. Gracias a la inmunización con nirsevimab, administrada a más del 90 % de los lactantes menores de seis meses en el año 2023, se observó una reducción de hasta el 80 % en los ingresos hospitalarios por bronquiolitis aguda. La bronquitis en edad pediátrica comprende cuadros agudos, predominantemente virales, y formas persistentes asociadas a infección bacteriana, entre las que destaca la bronquitis bacteriana prolongada o persistente (BBP). La bronquitis aguda se manifiesta fundamentalmente con tos y puede acompañarse de síntomas constitucionales leves y curso autolimitado; el diagnóstico es clínico y el manejo sintomático con medidas de soporte. En contraste, la BBP se define por tos productiva crónica de más de 3-4 semanas de evolución, con adecuada respuesta a un ciclo prolongado de antibiótico de amplio espectro. La falta de respuesta terapéutica obliga a considerar diagnósticos diferenciales como bronquiectasias. |
Key words: Acute bronchiolitis; Bronchitis in pediatrics; Respiratory syncytial virus; Monoclonal antibody.
Palabras clave: Bronquiolitis aguda; Bronquitis en pediatría; Virus respiratorio sincitial; Anticuerpo monoclonal.
Pediatr Integral 2026; XXX(1): 29 – 43
OBJECTIVES
• To recognize the importance of acute bronchiolitis (AB) as a frequent cause of illness and hospitalization in infants.
• To know the main causative agents and the clinical and epidemiological characteristics of AB.
• To identify the severity criteria, risk factors, and complications associated with this disease.
• To understand current guidelines on the use of drugs (bronchodilators, corticosteroids, antibiotics) and describe supportive measures (oxygen therapy, hydration, suctioning of secretions).
• Understanding prevention as the most relevant strategy for reducing the incidence and complications of AB. Recognizing nirsevimab as a major advance in the prevention of acute bronchiolitis caused by respiratory syncytial virus, as it offers longer-lasting protection with a single dose, making it easier to apply at the population level.
• To recognize the typical clinical manifestations of acute bronchitis and protracted bacterial bronchitis (PBB).
• To distinguish acute bronchitis from other causes of prolonged wet cough, such as asthma, bronchiectasis, or chronic respiratory infections.
• To distinguish when a wet cough is suggestive of PBB and when further investigation with additional tests is indicated.
Acute bronchiolitis and bronchitis in pediatrics
https://doi.org/10.63149/j.pedint.110
Acute bronchiolitis
Introduction
Acute bronchiolitis is a prevalent condition in children under two years of age. It is the leading cause of pediatric hospital admissions during the winter season, with respiratory syncytial virus (RSV) being the most frequent cause. All bronchiolitis treatment guidelines emphasize reducing the use of diagnostic tests and treatments that are not evidence-based. Management is based on supportive measures. Prevention is evolving thanks to the use of monoclonal antibodies that protect the most vulnerable infants during RSV season and have been shown to significantly reduce hospitalizations and severe cases of bronchiolitis.
Definition
Acute bronchiolitis (AB) can be defined as the first episode of respiratory distress due to involvement of the distal bronchi in a child under 2 years of age, which is preceded by catarrhal symptoms.
Bronchiolitis is the most common acute lower respiratory tract infection in children under one year of age, which causes high rates of consultation, hospitalization and high health costs(1).
The most distal airways, which have a diameter of less than 2 mm and lack cartilage in their walls, are called bronchioles. In these airways, RSV triggers inflammation and necrosis of the epithelial cells, which, along with edema and increased mucus production, leads to obstruction.
Due to the difficulty in defining AB, various criteria have been proposed; the best known are still the McConnochie criteria (1993), according to which bronchiolitis is defined as the first acute episode of wheezing preceded by a respiratory picture of viral origin (rhinorrhea, cough, tachypnea with or without fever) that affects children under two years of age and, preferably, within the first year(2).
However, these clinical criteria for diagnosis are not universally accepted and vary between countries and clinical practice guidelines (CPGs).
Etiology
The most common cause of bronchiolitis is viral infection, with the viruses responsible varying depending on the season and geographic area. The most frequently isolated viruses in bronchiolitis are RSV, followed by rhinovirus.
RSV is the most frequently detected virus, responsible for approximately 70% of cases. It is found worldwide and causes seasonal outbreaks. It is named for its tendency to form clusters of giant cells, called syncytia, in tissue cultures. It is a pleomorphic single-stranded RNA virus with two subgroups (A and B) that differ in virulence and seasonal patterns (Fig. 1). Infection does not confer complete and lasting immunity, so a patient can be reinfected and experience bacterial pneumonia (BP) several times(3,4).
Figure 1. Structure of respiratory syncytial virus (RSV).
Source: Jung HI, et al. Viruses. 2020; 12: 102.
The second most common virus is rhinovirus (RV), which causes the common cold. It circulates year-round but has a higher incidence (up to 80%) in spring and autumn. Of its three species, type A causes the most severe bronchiolitis. These cases typically affect older children, who attend daycare, and are more likely to have atopic dermatitis and a family history of asthma or atopy than infants with bronchiolitis caused by RSV. Parainfluenza virus type 3 is associated with bronchiolitis epidemics in spring and autumn. Types 1 and 2 are more frequently associated with laryngitis but can also cause bronchiolitis.
Less frequently, the following are causes of wheezing: adenovirus, metapneumovirus (with a winter distribution similar to RSV), influenza A and B viruses, coronaviruses (including SARS-CoV-2), human bocavirus 1, and enteroviruses. Viral coinfections were present in 48.3% of cases. Occasionally, Mycoplasma pneumoniae and Bordetella pertussis can cause wheezing episodes in infants(5-7).
Microbiology
RSV belongs to the genus Orthopneumovirus, family Pneumoviridae. There are two main antigenic groups, A and B, and multiple genotypes within the two groups(3,4). It consists of the viral genome (single-stranded RNA, with 10 genes encoding 11 proteins), the nucleocapsid, and a lipid envelope to which 3 membrane proteins are anchored:
• G glycoprotein: it is responsible for the adhesion of the virus to the cell membrane and the antigenic variability between groups A and B.
• F glycoprotein: responsible for the fusion, penetration, and destruction of cell membranes. It is the preferred target for the development of vaccines, monoclonal antibodies, and antivirals. It can be present in two different spatial conformations within the same virus: the pre-fusion (pre-F) conformation, which is filamentous and highly infectious; and the post-fusion (post-F) conformation, which is spherical and more resistant to antibodies (Fig. 2).
• SH protein: of unknown function.
Figure 2. Configuration of pre-F and post-F forms. Source: Ruckwardt TJ, et al. Immunity. 2019; 51: 429-42.
Epidemiology
AB primarily affects children under one year old, with peak incidence between 3 and 6 months, and is more common in males. In the Northern Hemisphere, epidemic outbreaks are most frequent between November and April. Humans are the reservoir in whom it is highly infectious. The virus enters the body through the nasal, conjunctival, and/or oral mucosa. Transmission can occur through direct contact or via respiratory secretions.
It is a disease that impacts all levels of healthcare, especially during epidemic periods. Spain leads Europe in terms of healthcare costs and primary care visits for RSV in children under 5 years of age. It primarily affects children under one year old; the peak incidence is between 3 and 6 months, with a predominance in males(3). In the Northern Hemisphere, epidemic outbreaks are more common between November and April, with a peak in January and February; in tropical climates, they are associated with the rainy season. It does not produce lasting immunity, and reinfections are frequent.
Before the COVID-19 pandemic, the epidemiology of bronchiolitis in Spain followed a predictable and recurring pattern. Around 100,000 episodes were recorded annually in children under 2 years of age, with peak incidence during the colder months. The COVID-19 pandemic caused an unprecedented disruption to the epidemiology of bronchiolitis. The public health measures implemented, such as lockdowns, social distancing, widespread mask use, and hand hygiene, drastically reduced the circulation of respiratory viruses. As a result, the incidence of bronchiolitis fell sharply. In the 2020-2021 season, the cumulative incidence of hospitalizations plummeted to 3.6 per 10,000 children, representing a 67% decrease in cases compared to the 2019-2020 season. As pandemic restrictions were lifted, bronchiolitis returned with force and atypical behavior. The usual transmission pattern, which culminates in a winter peak, was disrupted, with a high incidence of cases outside the typically epidemic months. For example, in 2021, a high incidence of cases was observed between April and October, with an unusual peak in June. The return of bronchiolitis was particularly noticeable in the 2021-2022 season, when the incidence increased to 65.7 per 10,000 children, although it did not reach pre-pandemic levels. In addition to the increased incidence, an increase in the severity of cases was also observed.
In Spain, in the years prior to 2023, the rate of hospital admissions for acute bronchiolitis ranged from 20 to 40 per 1,000 live births under 12 months of age. These admissions accounted for 18% of all pediatric hospitalizations. Thanks to immunization with nirsevimab, administered to more than 90% of infants under six months of age in 2023, a reduction of up to 80% in hospital admissions for acute bronchiolitis was observed, preventing between 7,510 and 10,213 hospitalizations during the season(4).
According to data from the Acute Respiratory Infection Surveillance System (SiVIRA) and the analysis of hospitalizations using the Minimum Basic Data Set (CMBD)(8), in Spain, during the 2023-24 and 2024-25 seasons, the total estimated number of hospitalizations due to RSV in Spain is 29,081 cases per season (Fig. 3).
Figure 3. Incidence rates of acute bronchiolitis/bronchitis in children under 5 years of age in Primary Care, by sex. Source: SiVIRA, 2023-24 season.
The average duration of the illness was 10 days, with hospital admissions in 16.8% of cases, and an average of 4 days per hospitalization, while 1.5% of the children were admitted to the PICU.
The reservoir is humans, in whom it has a high infectivity. Transmission can occur through direct contact, via respiratory secretions where the virus can survive for 6 to 12 hours, through inhalation of large infectious droplets (contagious up to two meters away), and also through indirect contact via fomites (especially the hands of caregivers). The virus is effectively inactivated by washing hands with soap and water or using alcohol-based hand sanitizer.
The virus enters the body through the nasal, conjunctival, and/or oral mucosa. Once inside the body, RSV replicates in the nasal epithelium, triggering a broad immune response. After an incubation period of 2 to 8 days, in approximately one-third of infected patients, the infection spreads to the lower respiratory tract, where the virus replicates in the epithelial cells of the bronchiolar mucosa, generating another immune response.
The infant is usually contagious for about a week. Epithelial regeneration begins after 3-4 days, and ciliary recovery does not occur until after 15 days.
Pathogeny
RSV is a highly infectious virus, but it is a fact that not all infected patients develop bronchiolitis, and even fewer develop severe bronchiolitis. Damage to epithelial cells and the ciliary system are essential factors in the pathogenesis of the disease. The onset and severity of bronchiolitis will depend on:
• The virus: RSV-A produces more severe disease in some series.
• The host himself: risk factors of AB(9) (Table I).
• The individual’s own immune response (lower expression of Th1 lymphocytes and higher expression of Th2 lymphocytes). This imbalance contributes to increased inflammation and mucus production in the bronchioles(3). It has also been associated with greater disease severity, more extensive lung damage, and an increased risk of developing asthma or recurrent wheezing in the future. This alteration has been observed in clinical studies, where children with severe RSV bronchiolitis have a higher IL-4/IFN-γ ratio, indicating a predominance of the Th2 response. Single nucleotide polymorphisms (SNPs) are variations in the genetic sequence that can modify the expression or function of proteins involved in the immune response. In various sequencing assays, SNPs have been discovered in cytokines, receptors, and signaling molecules in children with bronchiolitis. These SNPs influence the intensity of inflammation and the ability to control viral infection, thus demonstrating an individual’s susceptibility to developing severe forms of bronchiolitis.
Pathophysiology
AB is the result of the interaction of the viral infection with the child’s immune system, which reacts with an exaggerated immune response, causing inflammation and bronchiolar obstruction, the origin of severe respiratory impairment in young children.
The immune system responds to RSV infection by releasing proinflammatory cytokines (such as IL-6, IL-8, and TNF-α), which attract leukocytes to the site of infection and produce an infiltrate of lymphocytes, neutrophils, and monocytes, intensifying local inflammation(7). RSV selectively infects ciliated cells and produces variable inflammation of the terminal bronchioles, mucosal edema, necrosis of the respiratory epithelium, and mucus hypersecretion with mucus plugs that block the bronchioles. As a consequence of the variable involvement of the bronchioles, complete obstruction (atelectasis) can occur in some lung segments, while air trapping and hyperinflation (emphysema) can occur in others, resulting in a patchy pattern(7). Decreased elasticity and increased pulmonary vascular resistance lead to increased work of breathing and impaired gas exchange. Obstruction and ventilation/perfusion imbalance produce hypoxemia, polypnea and tachypnea and, in severe cases, respiratory acidosis.
Bronchospasm, although not the main pathogenic mechanism, can contribute to exacerbating respiratory distress.
Clinical manifestations
RSV infection in children almost always produces clinical symptoms and is generally self-limiting. After a brief incubation period with upper respiratory symptoms, the acute phase begins, with lower airway involvement, severe cough, and increased work of breathing (tachypnea, intercostal retractions, wheezing, etc.), which usually improves after 2-5 days. The cough may persist for up to 3-4 weeks.
RSV infection in children almost always produces clinical symptoms, which are generally self-limiting and vary in severity depending on the patient’s age, risk factors, and previous exposure to the virus. The diagnosis of AB is primarily clinical, based on findings from the medical history and physical examination.
The initial phase begins after a brief incubation period, with upper respiratory symptoms such as congestion, rhinorrhea, sneezing, and cough(5). Approximately 50% of children present with fever or a low-grade fever, usually not very high (<38.5ºC). Within 1 to 4 days, the established phase begins, with involvement of the lower airway. The cough becomes more persistent, accompanied by irritability, feeding difficulties, and signs of increased work of breathing, such as tachypnea, chest retractions, and flaring, with peak symptoms on days 5-7. Auscultation may reveal a mixture of wheezing, prolonged expiration, and fine crackles(7).
Apnea, present in up to 23% of high-risk groups, such as premature infants, may be the most striking symptom in patients under two months old, who may not present other clinical signs.
Cough is the predominant symptom. It is usually dry, in fits, paroxysmal, but it does not usually present with the inspiratory “whoop” typical of pertussis.
Respiratory distress, which increases in intensity, manifests as intercostal or supraclavicular retractions, use of abdominal muscles, and nasal flaring; it reaches its peak within 24-48 hours, at which point most hospital admissions occur. Respiratory rate generally decreases between the second and fifth day; however, auscultatory signs persist in some infants for a week or more.
The clinical course in the absence of risk factors is favorable. During the convalescent phase, recovery from respiratory symptoms is expected. The average time for cough resolution ranges from 1 to 2 weeks, although 20% of patients may experience symptoms for up to three weeks. Viral shedding can persist for up to 4 weeks, especially in very young or immunocompromised patients. If the cough lasts longer than 3 weeks, reassessment and consideration of other underlying conditions are necessary.
Diagnosis
A thorough medical history and physical examination are usually sufficient to establish the diagnosis and classify the severity of the disease. Routine additional tests are discouraged and indicated only in specific cases.
The combination of a history of cold-like symptoms, progressive respiratory distress, tachypnea, wheezing, and low oxygen saturation confirms the diagnosis. Additional tests, such as blood tests or X-rays, are not usually necessary in most cases and are reserved for specific situations or to rule out other pathologies(6,7):
1. Medical history: the typical presentation occurs in children under 2 years of age, with a peak incidence between 3 and 6 months, with the presence of catarrhal symptoms in the preceding days, followed by the onset of a cough that worsens and is associated with increased respiratory effort. The presence of personal risk factors and feeding refusal or difficulty are signs of severity. Factors that contribute to the diagnosis include: occurring during an epidemic period, frequent family history of previous respiratory infection, being the first episode, and ruling out other possible causes(7).
2. Physical examination: the physical examination is essential to confirm the diagnosis and assess the severity.
• General condition and level of consciousness: assess the general condition (reactive, irritable or lethargic).
• Inspection: we will find tachypnea and other signs of respiratory distress, such as intercostal, subcostal and suprasternal retractions, nasal flaring, expiratory grunting and cyanosis.
• On pulmonary auscultation, we find the presence of wheezing, crackles or rales and a prolonged expiration.
• Pulse oximetry: this is indicated to assess the severity of the illness at the onset of symptoms and to monitor its progression. It must be interpreted in conjunction with the patient’s clinical condition, and continuous monitoring is not recommended. Low oxygen saturation (generally below 90-92%) is an indicator of severity and often requires hospitalization and oxygen therapy.
3. Additional tests: current clinical practice guidelines, both national and international, advise against routine additional tests and they are only indicated in specific cases(5,7,10):
• Chest X-ray: it is not necessary for the diagnosis of typical bronchiolitis and does not alter the management of bronchiolitis. It may be requested if a complication is suspected, such as pneumonia, atelectasis, heart failure, atypical clinical presentation, or unfavorable evolution. Radiological findings are variable and nonspecific: hyperinflation, peribronchial thickening and patchy atelectasis with volume loss.
• Laboratory diagnosis of RSV is primarily performed through:
– Real-time PCR: it is the most sensitive and specific, detecting genetic material from the virus.
– Rapid antigen detection in nasal samples is used especially in pediatrics because of its speed.
– Viral cultures, slower and less sensitive.
Identifying the causative agent does not appear to modify patient management, and current clinical practice guidelines (CPGs) do not recommend its routine identification; it can be useful in cases of hospital admission for epidemiological purposes and for the management of hospitalized patients.
• Laboratory tests: these are not routinely recommended; they may be useful if there is doubt about the diagnosis, to assess hydration status, or to rule out a bacterial superinfection. We may request procalcitonin and/or C-reactive protein if there is high fever and suspicion of potentially serious bacterial infection.
• Urine test strip, sediment and urine culture: indicated in children under 2-3 months with bronchiolitis and persistent axillary temperature >39ºC, since urinary tract infection is the most frequent coinfection in infants with bronchiolitis.
• Blood gas analysis is not routinely indicated, except if there are signs of respiratory distress, apnea, or patients requiring admission to Intensive Care. Capillary blood gas analysis has a reliability similar to arterial blood gas analysis.
• Lung ultrasound: due to its safety, it will be increasingly found at the patient’s bedside. It could be incorporated into the scores for management, as it has shown a sensitivity and specificity of 100% and 82%, respectively, for predicting admission to PICU.
Differential diagnosis of wheezing in infants
When a child under two years of age presents with a dry cough and respiratory distress with or without wheezing, it is crucial to consider a broad spectrum of differential diagnoses(5-7), since wheezing, although characteristic of bronchiolitis, is not exclusive to this condition. Cough and wheezing in bronchiolitis can be difficult to distinguish from other entities such as(5):
• Common respiratory conditions: asthma attacks (especially in children over 6 months old with an atopic family or personal history), pneumonia, pertussis, and virus-induced wheezing.
• Obstructive airway diseases: foreign body obstruction, pulmonary malformations, vascular rings, laryngotracheomalacia, tracheal stenosis or bronchostenosis, and lymphadenopathy or tumor that may compress the airways.
• Chronic diseases: cystic fibrosis, bronchopulmonary dysplasia (in premature infants), immotile cilia syndrome, alpha-1 antitrypsin deficiency and immunodeficiencies.
• Non-respiratory conditions with respiratory manifestations: gastroesophageal reflux, swallowing disorders, and congenital heart disease and heart failure.
Severity assessment
A fundamental part of the diagnosis is to assess the severity of the clinical picture (Table II), since this determines whether the child needs hospitalization or can be managed at home.
Commonly used severity scales incorporate key clinical parameters, such as respiratory rate (RR), heart rate (HR), oxygen saturation (SpO2), the presence and degree of retractions, and the characteristics of wheezing or crackles(5,6,11,16). The pursuit of objectivity in a variable clinical picture is reflected in the effort to standardize the assessment. The adoption of severity scales allows for more precise communication among healthcare professionals, facilitates the monitoring of clinical evolution and treatment response, and is fundamental for clinical research, enabling patient comparisons and the evaluation of treatment efficacy(14). Different assessment scales can be used: modified Wood-Downes, the respiratory distress index or RDAI, the Sant Joan de Déu scale (Table III) or the simplest and most recently validated in children with AB, the modified Tal scale(9) (Table IV), which assesses respiratory rate, oxygen saturation, retractions and level of consciousness.
The Sant Joan de Déu scale(15) is specifically designed and validated for AB (ESBA). It cumulatively assesses reproducible clinical parameters: respiratory and heart rate according to age, respiratory effort (retractions, nasal flaring), auscultation of adventitious sounds (wheezing, crackles), and inspiratory/expiratory ratio. Scores range from 0 to 13: mild (0 to 4 points), moderate (5 to 9), and severe (10 to 13). It has high reliability and validity for predicting the need for admission to the PICU (sensitivity: 82% and specificity: 91%).
Treatment
The vast majority of bronchiolitis cases are mild to moderate and can be managed at home. Treatment for bronchiolitis in infants is primarily supportive. Current clinical practice guidelines advise against the routine use of medications such as bronchodilators, corticosteroids, or antibiotics, as their efficacy has not been demonstrated and they may have adverse effects.
The goal of treatment is to relieve symptoms and ensure adequate oxygenation and hydration of the infant.
Supportive treatment(5-7,9,11-13,16)
• Position: placing the infant in semi-seated position (with a slight elevation of the head of +30°) can facilitate breathing.
• Nasal irrigation and aspiration of secretions: the use of saline nasal drops is effective, safe, and non-irritating. Apply several drops to each nostril and then suction out the secretions with a nasal aspirator, especially before feedings and at bedtime. Deep nasal aspiration beyond the nasopharynx is not recommended.
• Air humidification: if the air in the room is dry, a humidifier or cool mist vaporizer can moisten the environment, which helps to loosen mucus and relieve coughs.
• Ambient temperature: keep the ambient temperature no higher than 20 °C.
• Pain and fever relief: for the treatment of fever or pain, medications such as acetaminophen or ibuprofen can be administered, following the dosage instructions.
• Avoid environmental irritants: exposure to tobacco smoke (secondhand smoke) worsens the symptoms of respiratory infections. It is also advisable to avoid crowded places.
• Environment: ensure a calm environment and avoid disturbing the infant with sudden movements. Dress the baby in comfortable, loose-fitting clothing, avoiding overdressing.
• Hydration and nutrition: it is essential to ensure the infant remains well hydrated. Fluids (breast milk or formula) should be offered more frequently, but in smaller amounts, to prevent the infant from becoming overly tired while feeding. Intravenous or nasogastric tube feeding is reserved for babies who cannot tolerate oral feeding.
• Oxygen therapy: oxygen therapy is a crucial supportive measure in the management of bronchiolitis when the infant’s oxygenation is compromised. The goal varies depending on the guidelines used, but generally, it is to maintain an oxygen saturation level (SpO2) ≥90-92%. It is the only medical intervention with proven efficacy. Nasal cannulas are the preferred initial method for oxygen administration. Supplemental oxygen should be administered via nasal cannula, mask, or, in more severe cases, high-flow oxygen therapy (HFNC) and continuous positive airway pressure (CPAP); these are used to decrease respiratory effort, improve gas exchange, and avoid endotracheal intubation(6,9,11).
• Respiratory physiotherapy: recent clinical guidelines advise against routine respiratory physiotherapy. Studies have not demonstrated a clear benefit, and in some cases, it has been associated with adverse effects such as vomiting. However, it may be considered in children with bronchiolitis who have other comorbidities and difficulty clearing secretions.
• Daycare attendance: the infant should not attend daycare until the symptoms have completely disappeared.
Drugs not routinely recommended
Treatment for bronchiolitis is based on supportive measures. Clinical guidelines and medical practice agree that the routine use of certain drugs is not recommended, as they have not demonstrated efficacy in the progression of the disease.
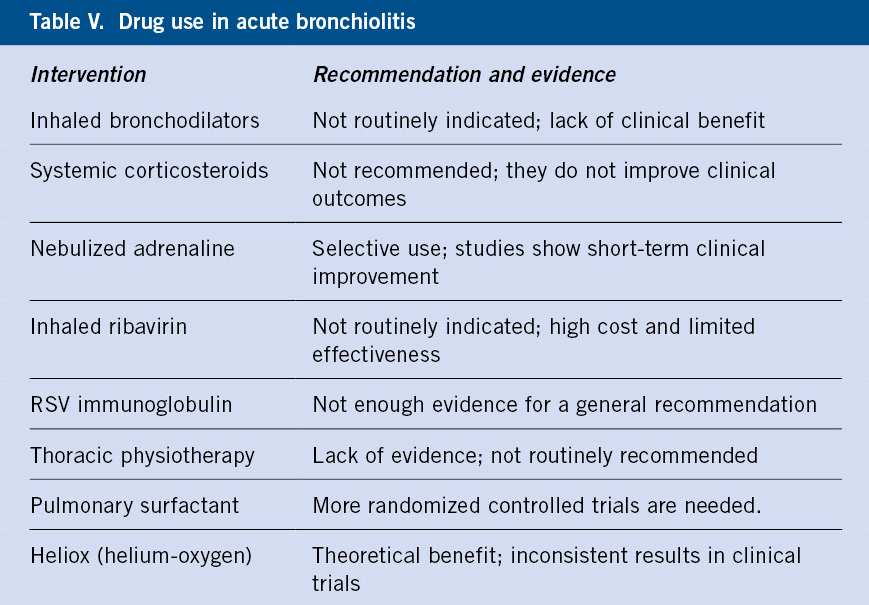
Current scientific evidence advises against(5-7,11-13,16) the use of the following medications (Table V):
• Bronchodilators (salbutamol, adrenaline): do not reduce the need for hospital admission or the duration of the illness in most cases. Their use is only considered in highly selected cases, as a therapeutic trial(9) in children older than 12 months with a personal (PH) or family (FF) history of atopy or asthma, outside the RSV epidemic period (November-February). If there is no clear and rapid response, administration should be discontinued. Nebulized adrenaline is not routinely recommended; there is no strong evidence that it reduces the number of admissions, or the length of hospital stay. Several studies conclude that there is insufficient evidence to recommend the routine use of nebulized adrenaline in acute bronchiolitis(5).
• Corticosteroids (oral, inhaled, or systemic): these have not been shown to be effective in improving short- or long-term outcomes of bronchiolitis and are not recommended. They also do not reduce the risk of recurrent wheezing in the future.
• Antibiotics: these are not indicated, as bronchiolitis is a viral illness. They should only be used if a bacterial superinfection is suspected.
• Ribavirin is the only antiviral with which there is some experience, although the available data are not sufficient to support its routine use.
• Hypertonic saline (3%): nebulization with hypertonic saline remains controversial. It should not be administered to children with bronchiolitis in the emergency department. Some research suggests it may reduce hospital stay in admitted patients, but the evidence is not yet strong enough to recommend its widespread use.
• Respiratory physiotherapy: it does not improve respiratory parameters, does not reduce the need for supplemental oxygen or the duration of the hospital stay.
Prevention
Prevention is a fundamental pillar of pediatric health and the best strategy for avoiding bronchiolitis. The most significant advance, and one that has changed the paradigm in the fight against RSV bronchiolitis, is the development of monoclonal antibodies.
The enormous impact of RSV infection and the lack of effective treatment make the development of vaccines and monoclonal antibodies against this virus a global priority.
General hygiene measures
Non-pharmacological prevention measures are crucial to limiting the spread of RSV and other respiratory viruses(5,7,11):
• Hand hygiene: frequent and proper hand washing with soap and water, or the use of hydroalcoholic hand sanitizer is the most effective measure to prevent the spread of the virus.
• Avoid contact with sick people: keeping the infant away from people with colds, coughs or fever is an essential measure to prevent contagion.
• Avoid exposure to tobacco smoke: smoking should be avoided inside the home or car.
• Environmental hygiene: frequently clean and disinfect toys and surfaces that may be contaminated.
• Ventilation: ventilate the rooms of the house at least once a day to keep the air clean.
• Breastfeeding: exclusive breastfeeding for at least 6 months strengthens the baby’s immune system and reduces the morbidity of respiratory infections.
• Daycare attendance: attendance at daycare is not recommended for children at high risk of severe bronchiolitis (children under 3 months, premature infants, or those with chronic illnesses).
Advances in prevention
The most significant advance, and one that has changed the model in the fight against RSV bronchiolitis, is the development of monoclonal antibodies (Table VI).
Currently, two monoclonal antibodies are approved for the passive prevention of RSV disease(4,17):
• Palivizumab (Synagys, 1998) is a humanized IgG1 monoclonal antibody produced by recombinant DNA technology in mouse myeloma host cells. It contains 95% human-identical genetic sequence and the remaining 5% is of murine origin. It acts by binding to the RSV F protein, inhibiting cell fusion. It has a half-life of 20 days. It is available in 0.5 ml (50 mg) and 1 ml (100 mg) vials.
• Nirsevimab (Beyfortus 2022) is a human immunoglobulin G1 kappa monoclonal antibody produced in Chinese hamster ovary cells using recombinant DNA technology. It is modified with a triple amino acid substitution (YTE) in the Fc region to extend its serum half-life, which ranges from 85 to 117 days. It inhibits the essential membrane fusion step in viral entry, neutralizing the virus and blocking cell-cell fusion. It is available in pre-filled syringes of 0.5 ml (50 mg) and 1 ml (100 mg). Unlike palivizumab, which required monthly injections throughout the viral season and was only indicated for high-risk infants, nirsevimab is a long-acting monoclonal antibody.
– A single dose: with a single injection at the start of RSV season (usually in autumn), the baby is protected for a period of up to five months.
– Broader reach: it is approved to protect all infants, not just those at high risk, representing a fundamental shift in disease prevention. Most cases of severe bronchiolitis occur in infants with no prior risk factors.
– It comes in a pre-filled syringe. It must not be shaken. No dilution is required. Available presentations are:
a. Beyfortus® 50 mg, for infants weighing less than 5 kg. In a pre-filled syringe with a purple plunger. It contains 50 mg of nirsevimab in 0.5 ml (100 mg/ml).
b. Beyfortus® 100 mg, for infants weighing 5 kg or more. In a pre-filled syringe with a light blue plunger. It contains 100 mg of nirsevimab in 1 ml (100 mg/ml).
Impact
The Public Health Commission recommended nirsevimab for infants under 6 months of age during RSV season and for high-risk groups(17). Coverage reached nearly 90% nationally, with early analyses estimating an 80-85% effectiveness rate against hospital admission in infants under 6 months of age (4). Acute RSV bronchiolitis remains the leading cause of pediatric hospitalization in infants in Spain.
Between 2023 and 2025, the widespread introduction of nirsevimab immunization and the start of maternal vaccination substantially reduced consultations and hospital admissions for bronchiolitis in infants. In the 2024-2025 season, the incidence of acute RSV bronchiolitis in children under 1 year of age was significantly lower than in pre-pandemic seasons(17) (Fig. 4).
Figure 4. Progression of the incidence of acute bronchiolitis in Spain (2019-2025).
A reduction in pediatric hospitalizations was observed, especially in immunized infants (Fig. 5).
Figure 5. Progression of pediatric hospitalizations due to RSV in Spain (children under 1 year old).
The peak of viral circulation remained in November and December, but with less pressure on healthcare services(4):
• Maternal vaccine against RSV(4): this vaccine is available in Spain under the name Abrysvo®. It is composed of a prefusion F protein antigen from subgroup A and another from subgroup B, produced in Chinese hamster ovary cells using recombinant DNA technology. It is indicated for pregnant women between weeks 32 and 36 of gestation. The mother develops antibodies that are transferred to the fetus through the placenta, protecting the baby for the first six months of life. If the mother is vaccinated during pregnancy, it is usually not necessary to administer nirsevimab to the newborn, unless the birth occurs out of season or there are additional risk factors. Its implementation in the RSV prevention strategy is under evaluation in Spain for the 2024-2025 season, although it is not yet publicly funded. It is presented in a box containing a vial of powder, a pre-filled syringe with the diluent, and a vial-to-syringe adapter. Other adjuvanted recombinant vaccines, vectored vaccines, and messenger RNA vaccines are also under development.
New treatments for AB
The future of AB treatment focuses on more specific therapies:
• Direct-acting antivirals: new antiviral drugs are being investigated to directly combat RSV. These drugs, which could be administered to hospitalized patients, aim to shorten the duration and severity of the infection.
• Inflammation management: given the inflammatory nature of the disease, new agents that can modulate the body’s immune response to reduce inflammation in the bronchioles are being investigated.
Prognosis and complications
Most cases of AB have a mild and self-limiting course, but complications such as apnea and bacterial infection can occur.
In most cases, acute bronchiolitis has a self-limiting course and a good prognosis, but some infants may develop severe forms that require hospitalization (Table VII) or even admission to an intensive care unit.
The most serious complications we can encounter are:
• Acute respiratory failure: which in severe cases may require hospitalization for ventilatory support treatment, with mechanical ventilation or CPAP. Some children develop recurrent wheezing or childhood asthma.
• Apnea, which may be the first and only sign of RSV infection and requires continuous monitoring. The incidence is higher in very premature infants, infants under 6 weeks old, or those with associated respiratory pathology.
• Bacterial superinfection (otitis or pneumonia) and dehydration are usually uncommon.
Bronchitis
Definition
Bronchitis or tracheobronchitis is defined as inflammation of the airways (bronchi associated or not with the trachea), usually caused by an infection, and is mainly manifested by coughing.
Bronchitis in pediatrics encompasses both acute, predominantly viral, forms and chronic forms, where prolonged bacterial infection is relevant and requires a specific diagnostic and therapeutic approach.
Acute bronchitis
In pediatrics, acute bronchitis is one of the most frequent respiratory infections, especially in children under 5 years of age, and is usually caused by viruses; because of this, it has a seasonal pattern, with cases increasing during the winter.
There are several infectious agents that cause acute bronchitis (Table VIII), with viral causes accounting for up to 90% of cases (adenovirus, influenza, parainfluenza, RSV, rhinovirus, bocavirus, coxsackievirus, herpes simplex). In patients with underlying lung disease or who are immunocompromised, it may be associated with bacteria (10% of cases), such as Streptococcus pneumoniae, Haemophilus influenzae, Moraxella catarrhalis, Mycoplasma pneumoniae, and Chlamydia pneumoniae. Rarely, it can be caused by other infectious agents, such as fungi, or non-infectious causes, such as allergies, aspiration, or gastroesophageal reflux.
Depending on the etiology, patients may present with general symptoms (malaise, nasal congestion, etc.) and fever, in addition to the typical clinical manifestation: a cough, which may be dry or productive, associated or not with respiratory distress, and occasionally, may present with abnormal auscultation findings such as wheezing or rhonchi. Up to 12.5% of patients may experience chest pain, which is more common in adolescents or pre-adolescents; this is usually benign and associated with intense coughing and inflammation of the respiratory mucosa, which stimulates pain receptors. Cases of chest pain without signs or symptoms suggestive of pneumonia, asthma, or cardiac pathology do not require further investigation(18-23).
The clinical course is self-limiting, and full recovery may take several weeks, 2-3 weeks, although in some cases it may take up to 4 weeks(24,25).
It is essential to differentiate acute bronchitis from other conditions, such as asthma or bronchiolitis, where bronchospasm and wheezing are the central characteristics.
The diagnosis is clinical, and rhonchi, wheezes, or crackles may sometimes be heard on auscultation. The decision to perform microbiological tests (PCR and multiplex panels) should be individualized and based on the likelihood that the result will modify clinical management. Since, in most cases, it will be a viral entity and management is symptomatic, routine complementary tests are not indicated(26,27).
Treatment (Table IX), in most cases, consists of symptomatic management with antipyretics, humidified environment, and general supportive measures. Occasionally, beta-2 bronchodilators are necessary in the presence of wheezing or bronchial hyperreactivity. Current evidence does not support the use of antitussives, honey, antihistamines, anticholinergics, oral nonsteroidal anti-inflammatory drugs (NSAIDs), or inhaled or oral corticosteroids. Antibiotics and antivirals should be considered in specific situations, according to scientific recommendations, but are not routinely necessary due to the risk of resistance. Hospital admission is exceptional.
Chronic bronchitis
The term chronic bronchitis in pediatrics is currently considered obsolete in the medical literature. The term was used to describe conditions characterized by a persistent wet cough secondary to recurrent inflammation of the bronchi, with increased secretions and/or impaired mucociliary clearance. In the last two decades, scientific evidence and clinical consensus have displaced this term in the pediatric population, favoring the use of persistent bacterial bronchitis (PBB) to define a chronic wet cough lasting more than four weeks that responds to antibiotic treatment and is not associated with other specific causes(26,27).
Therefore, according to current evidence, the use of PBB as a well-defined clinical entity is recommended and the use of chronic bronchitis in pediatrics is discouraged(26,27), since the latter is mainly related to adult pathology and does not adequately reflect the pathophysiology or the current therapeutic approach in children.
Persistent or protracted bacterial bronchitis (PBB)
Protracted bacterial bronchitis is characterized by a persistent productive cough lasting at least 3-4 weeks, with a good response to appropriate antibiotic treatment, and should not be accompanied by systemic symptoms or findings suggestive of alternative causes. Often, the cough worsens when lying down at night or upon waking in the morning and may be exacerbated by physical exercise. Therefore, it is sometimes difficult to distinguish between the cough of asthma and persistent bacterial bronchitis, and both can coexist in up to 30% of cases(29).
Although it can occur at any age, PBB usually affects children under 6 years old, and according to some studies, attending daycare or having a history of chronic cough may increase the risk of this condition. The main proposed cause is bacterial colonization of the respiratory tract, especially by Haemophilus influenzae, and the presence of concurrent viral infections (such as adenovirus), which would contribute to the pathogenesis of PBB(30).
Although the etiopathogenesis is unknown, it appears to be related to alterations in mucociliary clearance and inflammation caused by viral infections or environmental stimuli, such as smoke or air pollution. This, combined with possible bacterial superinfection and biofilm formation, would hinder eradication. These bacteria would produce an increase in inflammatory mediators, such as IL-8 and MMP-9, as well as neutrophilic inflammation, leading to an increase in purulent endobronchial secretions. In cases of prolonged or recurrent evolution, this inflammation could produce bronchial wall thickening, chronic neutrophilic inflammation, epithelial damage, and, in advanced cases, the development of bronchiectasis. In some cases, it can progress to chronic obstructive pulmonary disease (COPD) in adulthood, although this is less frequent in the pediatric population(29).
The main causative agents are non-typeable Haemophilus influenzae, Streptococcus pneumoniae, and, less frequently, Moraxella catarrhalis. Two or more pathogens are detected in half of the cultures.
Initially, when PBB was described in 2006, Chang et al. established diagnostic criteria (micro PBB) that included: chronic wet cough (more than 4 weeks), resolution with oral antibiotic therapy, and the need for microbiological isolation via bronchoscopy. In 2010, the definition of PBB was updated to facilitate its relevance in primary care, allowing diagnosis without the need for bronchoscopy for microbiological confirmation, prioritizing antibiotic response and the exclusion of other causes of wet cough (clinical PBB).
Treatment is based on a broad-spectrum antibiotic administered for four weeks, given the frequent polymicrobial etiology. The most commonly used regimen is amoxicillin-clavulanic acid 80-90 mg/kg/day every eight hours orally, and macrolides in cases of immediate hypersensitivity to penicillin. A first evaluation is recommended at two weeks, and the response should be assessed before extending antibiotic therapy to complete the four-week course. However, relapses are frequent (up to 25%) and require further courses of antibiotic therapy.
In those patients who do not show clinical improvement of symptoms or recurrence of the episode, different differential diagnoses should be considered; for this, the study should be expanded with a blood count and biochemistry with quantification of immunoglobulins, sweat test and, sometimes, high resolution computed axial tomography (HRCT), bronchiectasis is observed in up to 83.3% of cases of chronic wet cough that does not improve with adequate antibiotic treatment(31).
It is essential to rule out other causes of chronic cough, such as asthma or bronchiectasis, in case of recurrence or lack of response to treatment.
Similarities have been found between PBB, bronchial suppurative lung disease (BSLD), and bronchiectasis (BE). It is hypothesized that these may represent the same entity at different stages of chronic bronchial wall damage, with PBB being the mildest and most frequent, and bronchiectasis the most severe. Bronchiectasis is defined as irreversible bronchial dilatations visible on radiology. Patients with BE present with a persistent wet cough that responds partially or not at all to antibiotic treatment. In contrast, BSLD is used when a child presents with symptoms consistent with bronchiectasis—that is, recurrent or prolonged episodes of chronic wet cough—along with other symptoms such as failure to thrive, clubbing, abnormal chest sounds, and air trapping, but without radiological evidence of bronchiectasis.
Possible clinical progression: persistent bacterial bronchitis → chronic suppurative lung disease → bronchiectasis(31).
Role of the Primary Care Pediatrician
The primary care pediatrician serves as the first point of contact for the diagnosis, monitoring, and treatment of acute bronchiolitis. Therefore, the pediatrician’s role is crucial for early diagnosis, close monitoring of the illness to identify potential complications early and addressing families’ questions about the disease’s progression and duration. Accurately assessing the severity of the condition is another essential task for the pediatrician, key to deciding the child’s care: home if the case is mild, or hospital if there are warning signs or significant risk factors. Avoiding unnecessary treatments (bronchodilators, corticosteroids, antibiotics) and ensuring the safety of infants are also responsibilities of the primary care pediatrician. Supportive measures—adequate hydration, fever control, and nasal suctioning—are the priorities for keeping the infant comfortable. Finally, the pediatrician must strive to implement effective preventive measures: breastfeeding, hand hygiene, avoiding exposure to tobacco smoke, limiting contact with people with respiratory infections, and providing both vaccines and monoclonal antibodies, as a fundamental strategy to prevent bronchiolitis.
The primary care pediatrician also plays an essential role in the diagnosis and initial management of acute bronchitis in childhood. Their main function is to recognize acute, predominantly viral, and self-limiting cases, avoiding unnecessary tests and empirical antibiotic treatments that promote resistance. They must also identify persistent cases of productive cough suggestive of primary bronchitis, assess the response to antibiotic treatment, rule out differential diagnoses such as asthma or bronchiectasis in the absence of improvement, and make timely referrals if the situation requires it.
Conflict of interest
There is no conflict of interest in the preparation of this manuscript nor any source of funding.
References
The asterisks indicate the article’s level of interest, in the authors’ opinion.
1. Benito Fernández J, Paniagua Calzón N. Protocolos diagnósticos y terapéuticos en urgencias de Pediatría. Sociedad Española de Urgencias Pediátricas (SEUP). Diagnostic and therapeutic protocols in pediatric emergencies. Spanish Society of Pediatric Emergencies (SEUP). 3rd ed. 2019. Available at: https://seup.org/wp-content/uploads/2024/04/5_Bronquiolitis_4ed.pdf.
2. McConnochie KM. Bronchiolitis: What’s in the name? Am J Dis Child. 1993; 137: 11-3.
3.* Ridao Redondo M. Bronquiolitis y bronquitis. Bronchiolitis and bronchitis. Pediatr Integral. 2021; 1: 21-8. Available at: https://pediatriaintegral.es/wp-content/uploads/2021/xxv01/03/n1-021-028_MarisaRidao.pdf.
4.* Comité Asesor de Vacunas e Inmunizaciones de la AEP. Virus respiratorio sincitial. Cap. 43. En: Manual de inmunizaciones en línea de la AEP. Spanish Association of Pediatrics (AEP) Advisory Committee on Vaccines and Immunizations. Respiratory Syncytial virus. Chapter 43. In: AEP Online Immunization Manual. Available at: https://vacunasaep.org/documentos/manual/cap-43%2313.
5.** Duelo Marcos M, García Merino A, Mora Gandarillas I. Bronquiolitis Aguda. Protocolos del GVR (publicación P-GVR-4). Acute Bronchiolitis. GVR Protocols (publication P-GVR-4). Available at: https://www.respirar.org/index.php/protocolos.
6. Pérez Sanz J. Bronquitis y bronquiolitis. Bronchitis and bronchiolitis. Pediatr Integral. 2016; 1: 28-37. Available at: https://www.pediatriaintegral.es/publicacion-2016-01/2016-bronquitis-y-bronquiolitis/.
7. García García ML, Korta Murua J, Callejón Callejón A. Bronquiolitis aguda viral. Acute viral bronchiolitis. Protoc diagn ter pediatr. 2017; 1: 85-102.
8. Instituto de Salud Carlos III. Vigilancia de Infección Respiratoria Aguda en Atención Primaria y Hospitales. Gripe, COVID-19 y VRS. Carlos III Health Institute. Surveillance of Acute Respiratory Infection in Primary Care and Hospitals. Influenza, COVID-19 and RSV. In: Annual Report Season 2023-2024. Available at: https://cne.isciii.es/es/servicios/enfermedades-transmisibles/enfermedades-a-z/gripe-covid-19-y-otros-virus%20respiratorios.
9. Benito Fernández J, Paniagua Calzón N. Diagnóstico y tratamiento de la bronquiolitis aguda en Urgencias. Diagnosis and treatment of acute bronchiolitis in the Emergency Department.Protoc diagn ter pediatr. 2020; 1: 63-73. Available at: https://static.aeped.es/05_bronquiolitis_26d6c3bae7.pdf.
10. De la Flor i Brú J, Marès Bermúdez J; Grupo TECDIAP. Decálogo para la utilización del diagnóstico etiológico de bronquiolitis en Atención Primaria. Ten guidelines for the use of etiological diagnosis of bronchiolitis in Primary Care. Pediatr Integral. 2022; 5: 319-20. Available at: https://www.pediatriaintegral.es/wp-content/uploads/2022/xxvi05/10/n5-319-320_Int-esp_J-Flor.pdf.
11. Guía de Práctica Clínica sobre la Bronquiolitis Aguda. Plan de Calidad para el Sistema Nacional de Salud (SNS) del Ministerio de Sanidad y Política Social; 2010. Clinical Practice Guideline on Acute Bronchiolitis. Quality Plan for the National Health System (NHS) of the Ministry of Health and Social Policy; 2010. Available at: https://seup.org/pdf_public/gt/respiratoria_g_bronquio.pdf.
12.* González Hermosa A, Rodríguez Albarrán I. Bronquiolitis aguda en Urgencias de Pediatría. Exámenes complementarios y tratamiento. Revisión de la literatura (I) Acute bronchiolitis in the Pediatric Emergency Department. Complementary examinations and treatment. Literature review (I) Emerg Pediatr. 2022; 1: 22-9. Available at: https://seup.org/pdf_public/Revista_EP/R1_22_29_22.pdf.
13.* González Hermosa A, Llera Estefanía M, Montejo Fernández M. Bronquiolitis aguda en Urgencias de Pediatría. Exámenes complementarios y tratamiento. Revisión de la literatura (II). Acute bronchiolitis in the Pediatric Emergency Department. Complementary examinations and treatment. Literature review (II). Emerg Pediatr. 2022; 1: 83-92. Available at: https://seup.org/pdf_public/Revista_EP/R2_83_92_22.pdf.
14. Ramos-Fernández JM, Piñero-Domínguez P, Abollo-López P, Moreno-Pérez D, Cordón-Martínez AM, Milano-Manso G, et al. Estudio de validez de una escala de gravedad de la bronquiolitis aguda para orientar el ingreso en UCIP. Validity study of an acute bronchiolitis severity scale to guide admission to the PICU. Anales de Pediatría. 2018; 89: 104-10. Available at: https://doi.org/10.1016/j.anpedi.2017.09.006.
15. Balaguer M. Bronchiolitis: Score of Sant Joan de Deu: BROSJOD Score, Validation and usefulness. Pediatr Pulmonol. 2017; 52: 533-39. Available at: https://doi.org/10.1002/ppul.23546.
16. Claudia Fuentes S, Guillermo Cornejo C, Raúl Bustos B. Actualización en el tratamiento de bronquiolitis aguda: menos es más. Update on the treatment of acute bronchiolitis: less is more. Neumol Pediatr. 2016; 11:65-70.
17.** Grupo de trabajo inmunización frente a VRS población infantil de la Ponencia de Programa y Registro de Vacunaciones. Recomendaciones de inmunización pasiva para prevención de enfermedad grave por VRS en la población infantil. Comisión de Salud Pública del Consejo Interterritorial del Sistema Nacional de Salud. Ministerio de Sanidad. Working Group on RSV Immunization in Children, Program and Registry of Vaccinations Committee. Recommendations for passive immunization for the prevention of severe RSV disease in children. Public Health Commission of the Interterritorial Council of the National Health System. Ministry of Health. 2024. Available at: https://www.sanidad.gob.es/areas/promocionPrevencion/vacunaciones/VRS/poblacionInfantil/docs/VRS_infantil.pdf.
18.* Albert RH. Diagnosis and treatment of acute bronchitis. Am Fam Physician. 2010; 82: 1345-50. Available at: https://www.aafp.org/pubs/afp/issues/2010/1201/p1345.html.
19. Albi Rodríguez MS, Reig Rincón de Arellano I. Bronquitis (traqueobronquitis) aguda. 2024. Guía-ABE. Infecciones en Pediatría. Guía rápida para la selección del tratamiento antimicrobiano empírico. Acute Bronchitis (Tracheobronchitis). 2024. ABE Guide. Infections in Pediatrics. Quick guide for the selection of empirical antimicrobial treatment. Available at: https://www.guia-abe.es/temas-clinicos-bronquitis-(traqueobronquitis)-aguda.
20. Driscoll DJ, Glicklich LB, Gallen WJ. Chest pain in children: a prospective study. Pediatrics. 1976; 57: 648-51. Available at: https://doi.org/10.1542/peds.57.5.648.
21. Wenzel RP, Fowler AA. Clinical practice. Acute bronchitis. N Engl J Med. 2006; 355: 2125-30. Available at: https://doi.org/10.1056/NEJMcp061493.
22. Kinkade S, Long NA. Acute Bronchitis. Am Fam Physician. 2016; 94: 560-5. Available at: https://www.aafp.org/pubs/afp/issues/2016/1001/p560.html.
23. Okyere D, Nehme E, Mahony E, Stub D, Dawson LP, Ball J, et al. Incidence, diagnoses, and outcomes of pediatric nontraumatic chest pain attended by ambulance. JAMA Netw Open. 2025; 8: e2533962. Available at: https://doi.org/10.1001/jamanetworkopen.2025.33962.
24.** Mulhem E, Patalinghug E, Eraqi H. Acute bronchitis: Rapid evidence review. Am Fam Physician. 2025; 111: 214-7.
25. Thompson M, Vodicka TA, Blair PS, Buckley DI, Heneghan C, Hay AD, et al. Duration of symptoms of respiratory tract infections in children: systematic review. BMJ. 2013; 347: f7027. Available at: https://doi.org/10.1136/bmj.f7027.
26. Miller JM, Binnicker MJ, Campbell S, Carroll KC, Chapin KC, González MD, et al. Guide to utilization of the microbiology laboratory for diagnosis of infectious diseases: 2024 update by the Infectious Diseases Society of America (IDSA) and the American Society for Microbiology (ASM). Clin Infect Dis. 2024. Available at: https://doi.org/10.1093/cid/ciae104.
27. Caldwell JM, Espinosa CM, Banerjee R, Domachowske JB. Rapid diagnosis of acute pediatric respiratory infections with point-of-care and multiplex molecular testing. Infection. 2025; 53: 1-14. Available at: https://doi.org/10.1007/s15010-025-02553-5.
28.** Zhang J, Wurzel DF, Perret JL, Lodge CJ, Walters EH, Dharmage SC. Chronic bronchitis in children and adults: Definitions, pathophysiology, prevalence, risk factors, and consequences. J Clin Med. 2024; 13: 2413. Available at: https://doi.org/10.3390/jcm13082413.
29. Fernández Ventureira V, García Vera C. Bronquitis bacteriana persistente, una entidad a considerar en pediatría. Persistent bacterial bronchitis, an entity to consider in pediatrics. Anales de Pediatría. 2020; 93: 413-5. Available at: https://doi.org/10.1016/j.anpedi.2020.01.015.
30. O’Grady K-AF, Mahon J, Arnold D, Grimwood K, Hall KK, Goyal V, et al. Predictors of the development of protracted bacterial bronchitis following presentation to healthcare for an acute respiratory illness with cough: Analysis of three cohort studies. J Clin Med. 2021; 10: 5735. Available at: https://doi.org/10.3390/jcm10245735.
31.** Busquets Monge RM, Castillo Corullón S, Aguilar Fernández AJ. Tos húmeda: bronquitis bacteriana persistente, enfermedad supurativa bronquial y bronquiectasias. Protoc diagn ter pediatr. 2017; 1: 15-29. Wet cough: persistent bacterial bronchitis, suppurative bronchial disease, and bronchiectasis. Protoc diagn ter pediatr. 2017; 1: 15-29. Available at: https://static.aeped.es/02_tos_humeda_13c9f90ce5.pdf.
32. Ballesteros A, Crespo MP. Bronquitis bacteriana prolongada: luces y sombras. AEPap.org. Prolonged bacterial bronchitis: lights and shadows. AEPap.org. Available at: https://aepap.org/wp-content/uploads/2024/02/Bronquitis-bacteriana-prolongada-luces-y-sombras.-GPI-2017.pdf.
Recommended bibliography
– Duelo Marcos M, García Merino A, Mora Gandarillas I. Bronquiolitis Aguda. Protocolos del GVR (publicación P-GVR-4). Acute Bronchiolitis. GVR Protocols (publication P-GVR-4). Available at: https://www.respirar.org/index.php/protocolos.
Periodically reviewed technical protocols, geared towards the clinical practice of pediatricians, addressing the management of acute bronchiolitis, with key points being supportive measures and prevention.
– Comité Asesor de Vacunas e Inmunizaciones de la AEP. Virus respiratorio sincitial. Cap. 43. En: Manual de inmunizaciones en línea de la AEP. Spanish Association of Pediatrics (AEP) Advisory Committee on Vaccines and Immunizations. Respiratory Syncytial virus. Chapter 43. In: AEP Online Immunization Manual. Available at: https://vacunasaep.org/documentos/manual/cap-43%2313.
A very comprehensive document, updated on 2025, which reviews practical issues regarding the epidemiology, pathophysiology and, fundamentally, the immunization strategies against RSV.
– Grupo de trabajo inmunización frente a VRS población infantil de la Ponencia de Programa y Registro de Vacunaciones. Recomendaciones de inmunización pasiva para prevención de enfermedad grave por VRS en la población infantil. Comisión de Salud Pública del Consejo Interterritorial del Sistema Nacional de Salud. Ministerio de Sanidad. Working group on RSV immunization in the child population of the Immunization Program and Registry Committee. Recommendations on passive vaccination for the prevention of severe RSV disease in children. Public Health Commission of the Interterritorial Council of the National Health System. Ministry of Health. 2024. Available at: https://www.sanidad.gob.es/areas/promocionPrevencion/vacunaciones/VRS/poblacionInfantil/docs/VRS_infantil.pdf.
This is a key document because it establishes the official recommendations in Spain on passive immunization to prevent severe RSV disease in infants and young children. It marks a change in the model: from selective prevention (palivizumab in at-risk groups) to universal prevention with nirsevimab to prevent severe bronchiolitis in infants and reduce hospitalizations and serious RSV complications.
– Albert RH. Diagnosis and treatment of acute bronchitis. Am Fam Physician. 2010; 82: 1345-50. Available at: https://www.aafp.org/pubs/afp/issues/2010/1201/p1345.html.
It provides a clinical review on the diagnosis and management of acute bronchitis, including aspects relevant to the pediatric population.
– Mulhem E, Patalinghug E, Eraqi H. Acute bronchitis: Rapid evidence review. Am Fam Physician. 2025; 111: 214-7.
The text emphasizes that acute bronchitis is a self-limiting illness, predominantly viral in origin, and therefore antibiotics are not indicated in most pediatric cases. It underscores that there is no evidence to support the use of antitussives, honey, antihistamines, anticholinergics, oral NSAIDs, or inhaled or oral corticosteroids in the treatment of acute bronchitis in children.
– Zhang J, Wurzel DF, Perret JL, Lodge CJ, Walters EH, Dharmage SC. Chronic bronchitis in children and adults: Definitions, pathophysiology, prevalence, risk factors, and consequences. J Clin Med. 2024; 13: 2413. Available at: https://doi.org/10.3390/jcm13082413.
I consider this the most important article on the topic of prolonged bacterial bronchitis in pediatrics, because it provides a comprehensive and up-to-date review of the definition, pathophysiology, risk factors, and consequences of chronic bronchitis in children, specifically including the concept of prolonged bacterial bronchitis (PBB) as a distinct pediatric entity.
– Busquets Monge RM, Castillo Corullón S, Aguilar Fernández AJ. Tos húmeda: bronquitis bacteriana persistente, enfermedad supurativa bronquial y bronquiectasias. Wet cough: persistent bacterial bronchitis, suppurative bronchial disease, and bronchiectasis. Protoc diagn ter pediatr. 2017; 1: 15-29. Available at: https://static.aeped.es/02_tos_humeda_13c9f90ce5.pdf.
The periodically reviewed protocols establish an evidence-based clinical and therapeutic framework for the management of persistent bacterial bronchitis, suppurative bronchial disease, and bronchiectasis in pediatrics.
| Clinical case |
|
Patient data: age: 4 months; Sex: Female; Weight: 6.2 kg; Medical history: Full-term pregnancy, without complications. No family history of asthma or allergies. No RSV vaccination. Attending daycare for 2 months. Reason for consultation: the child is brought to the health center because her mother reports that for the past 48 hours, the girl has had a dry cough, nasal congestion, and difficulty feeding. Over the last 12 hours, the mother has noticed progressive respiratory distress with a dry cough, rapid breathing, wheezing, and a fever of 38.5°C. The mother also reports that the girl has been less hungry and more lethargic than usual. Examination: temperature: 38°C; respiratory rate: 60 rpm; heart rate: 150 bpm; oxygen saturation: 92% on room air; pulmonary auscultation: bilateral wheezing, fine crackles, subcostal retractions; general condition: irritable, with difficulty sucking. Additional studies: PCR for respiratory viruses: positive for RSV. Diagnosis: mild-to-moderate acute bronchiolitis due to RSV in an infant under 6 months of age. The parents were instructed to manage the child on an outpatient basis with home monitoring: paracetamol for fever, aspiration of nasal secretions, semi-upright position, fractionation of feeds to facilitate feeding and clinical follow-up in 24-48 hours.
|
 Acute bronchiolitis and bronchitis in pediatrics
Acute bronchiolitis and bronchitis in pediatrics