|
| Topics on Continuous Training |
D. Aguilera Alonso*, A. Lowy Benoliel**
*Infectious Diseases Section, Pediatric Service, Gregorio Marañón Health Research Institute (IiSGM), HGU Gregorio Marañón. Madrid. CIBER for Infectious Diseases (CIBERINFEC), Carlos III Health Institute. Madrid. **Otorhinolaryngology Department, HGU Gregorio Marañón. Madrid
| Abstract
The salivary glands, responsible for the production of saliva, have a fundamental role in digestion and phonation, and also have defense functions in the oral cavity. The largest glands, the parotid glands, are the most frequently affected. Salivary gland pathology includes several diseases of different aetiologies, including infectious, inflammatory, obstructive, autoimmune, granulomatous or neoplastic. In children, the most common disease of the salivary glands is epidemic parotitis, also known as mumps, followed by juvenile recurrent parotitis. |
| Resumen
Las glándulas salivales, encargadas de la producción de saliva, tienen un papel fundamental en la digestión y fonación y, además, tiene funciones de defensa en la cavidad oral. Las glándulas de mayor tamaño, las parótidas, son las más frecuentemente afectadas. La patología de las glándulas salivales engloba a un conjunto de enfermedades de etiologías diferentes, que incluye procesos: infecciosos, inflamatorios, obstructivos, autoinmunes, granulomatosos o neoplásicos. En niños, la enfermedad más frecuente de las glándulas salivales es la parotiditis epidémica, también conocida como paperas, seguida por la parotiditis recurrente de la infancia. |
Key words: Salivary glands; Parotitis; Mumps; Pediatrics; Epidemiology.
Palabras clave: Glándulas salivales; Parotiditis; Paperas; Pediatría; Epidemiología.
Pediatr Integral 2022; XXVI (7): 423 – 433
OBJECTIVES
• To recognize the main pathologies that affect the salivary glands.
• To establish the differential diagnosis in the care of a child with parotid tumor.
• To guide the diagnostic and therapeutic approach of recurrent parotitis.
• To be aware of the main tumors of the salivary glands.
 |
 |
Salivary gland pathology
Introduction
In the human species there are two types of salivary glands: major and minor(1). The major salivary glands are constituted by: two parotid glands, two submandibular or submaxillary glands, and two sublingual glands. The minor salivary glands are distributed throughout the mouth, pharynx, and paranasal sinuses. The main function of the salivary glands is the production of saliva. Saliva is of paramount importance for all the functions for which the oral cavity is responsible. It contains enzymes like amylase and lipase, which help in the digestion of food. In addition, they help in the sense of taste and in the lubrication of the oral cavity, as well as in the correct articulation and modulation of speech. It also has antimicrobial properties, as it contains secretory IgA immunoglobulins, mucins, and lactoferrin.
The largest salivary gland is the parotid gland, which produces a serous secretion that drains into the oral cavity through Stenon’s duct, at the level of the upper second molar. The submandibular gland produces a seromucous secretion that exits through Wharton’s duct into the floor of the mouth, while the sublingual gland produces a seromucous secretion that reaches the anterior region of the floor of the mouth.
Salivary gland pathology encompasses a set of diseases of different etiologies, including processes of: inflammatory, infectious, obstructive, autoimmune, granulomatous or neoplastic nature(2). In children, the most common disease of the salivary glands is epidemic parotitis, followed by recurrent parotitis of childhood.
Infectious sialadenitis
Sialadenitis is the name given to the inflammation of the salivary glands, with inflammation of the parotid gland (parotitis) being the most frequent presentation.
Epidemic parotitis (mumps)
Acute infection that typically produces inflammation of the parotid glands, predominantly bilateral, caused by the mumps virus. Other organs, such as: testicles, pancreas, ovaries and central nervous system, may also be affected simultaneously or in isolation.
The mumps virus is a member of the Paramyxoviridae family. It is a single-stranded RNA virus, for which humans are the only known natural host. Of its structural proteins, F and HN proteins are the main determinants of immunity. Although only one serotype of mumps virus is known, 13 genotypes have been determined(3).
Epidemiology
Transmission of the virus occurs by droplet spread from the respiratory tract and by direct contact with the saliva of an infected person. The distribution is universal, with a higher incidence in winter and spring(4). Since the introduction of the vaccine in Spain in 1981, its incidence has drastically fallen. However, epidemic waves have persisted every 3 to 5 years, mainly in people not immunized by the vaccine and, occasionally, also among immunized people, probably related to a loss of vaccine immunity over time. Currently, in Europe, G1 is the predominant circulating genotype(5).
Traditionally, the infection mainly took place between the ages of 5 and 14. In recent decades, due to systematic vaccination, it has shifted towards adolescents and young adults. Most of the reported cases of mumps in Spain correspond to individuals over 15 years of age, unvaccinated or vaccinated between 1993 and 1999 with a triple viral vaccine, whose anti-mumps component, the Rubini strain, was poorly immunogenic. Children under one year of age rarely acquire the infection, since they are protected by transplacental maternal antibodies.
The period of maximum contagion occurs from 2 days before to 5 days after the appearance of the parotid swelling, although the mumps virus has been isolated in saliva up to 9 days after the appearance of the parotiditis. Viral replication in the nasopharyngeal mucosa and regional lymph nodes is followed by a viremic phase, which causes spread of the infection to multiple organs, including the central nervous system and glandular epithelium.
It is a notifiable disease. The Epidemiological Surveillance Network classifies cases of mumps into:
• Suspected case: person who meets the clinical criteria (Table I).
• Probable case: person who meets the clinical criteria and the epidemiological criteria (Table I).
• Confirmed case: recently unvaccinated person (within six weeks prior to onset of symptoms) who meets clinical and laboratory criteria (Table I). A recently vaccinated person in whom the wild-type genotype of the virus is detected.
In addition, an outbreak is defined as: the appearance of two or more related cases.
Manifestations
The incubation period is on average 16-18 days, with an interval of 2-4 weeks. One-third of patients with mumps have subclinical disease or mild respiratory symptoms. Cases of “breakthrough” mumps in vaccinated individuals seem to have a more latent clinical course. As in other infections, its presentation in adults is usually associated with greater severity.
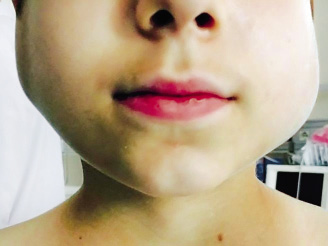
The most common manifestation is painful swelling of the parotid gland (present in around 95% of symptomatic patients), which is usually unilateral at the onset of the disease, and affects both parotid glands in the following days in 70-80% of cases (Fig. 1)(3).
Figure 1. 10-year-old boy with epidemic parotitis.
Prodromal symptoms, including fever, malaise, headache, and anorexia, usually precede mumps within the first few hours or days. At that time, ear pain ipsilateral to the swollen parotid gland and discomfort when eating or drinking acidic foods are common.
The swelling reaches its maximum size in about 3 days, stays that way for 1-2 days, and then slowly subsides. The orifice of Stenon’s duct usually appears edematous and erythematous. Occasionally, a morbilliform rash appears. Involvement of the other salivary glands may be seen in up to 10% of cases. Trismus may occur from mumps, and the patient may have difficulty speaking and chewing. The intake of citrus fruits or juices usually worsens the pain, which is very characteristic. As it obscures the mandibular angle, causing in some cases, elevation of the earlobe, allows to differentiate it from other cervical tumors, such as adenitis(4).
Complications
Central nervous system involvement is the most common extrasalivary manifestation. There is pleocytosis, with lymphocyte predominance in 50% of cases, without associating other data of meningitis. Clinical meningitis occurs, more frequently in males, in 1-10% of cases, generally several days after parotid swelling, but can occur a few days before or up to two weeks later, and may even not be associated with parotitis. The virus can be isolated in culture or detected by other microbiological techniques in cerebrospinal fluid (CSF). The presentation includes typical signs and symptoms of viral meningitis (headache, vomiting, fever and stiff neck). The CSF has 10 to 2,000 leukocytes/mm3, predominantly lymphocytes, although up to 25% may have a predominance of polymorphonuclear cells, mainly at the beginning. Proteinorrhachia is normal or slightly elevated. Hypoglycorrhachia has been described in up to 30% of patients. Meningitis is benign, with complete recovery and no sequelae.
Encephalitis has been described in 1 in 6,000 cases of mumps. It can appear both at the beginning of the disease (associated with direct damage due to invasion of the brain parenchyma by the virus), or late, 7-10 days after the onset of mumps (associated with a post-infectious demyelinating process produced by an autoimmune response). The cytochemical characteristics of the cerebrospinal fluid are similar to the meningitis described above. Patients usually present marked changes in the level of consciousness, associated with seizures, paresis, aphasia, and involuntary movements. Sequelae have been described, such as psychomotor retardation and seizure disorders, with a mortality of around 1%.
Other neurological complications that have been described are: Guillain-Barré syndrome, cerebellar ataxia, transverse myelitis and facial paralysis. The auditory nerve can also be affected, causing hearing loss.
Orchiepididymitis is the most frequent extrasalival manifestation in adults, present in 20-30% of postpubertal adolescent males with mumps. It is very rare before puberty. It is bilateral in one out of six patients and generally occurs during the first week of mumps. Some degree of atrophy has been described in half of the cases, but sterility is very rare. Oophoritis occurs in 5% of postpubertal women with mumps.
Around 3% of patients may present signs suggestive of pancreatitis, especially in older children and young adults; usually in the form of a mild and self-limited illness. In mumps virus infections, it is common to observe an increase in serum amylase of salivary origin, so serum lipase will be useful to differentiate this complication.
Other less frequent complications may appear, such as: diabetes mellitus; myocarditis, which is also usually mild and transient, but which, in rare cases, can be fatal; presternal edema due to obstruction of the lymphatic drainage of the neck; sialectasis with sialadenitis; arthritis; thyroiditis; nephritis; hepatitis; mastitis; prostatitis; optic neuritis; hemolytic anemia; or thrombocytopenia.
Infection during pregnancy poses a risk of miscarriage in the first trimester, although maternal-fetal transmission has not been described nor is there a clear risk of congenital malformations.
Differential diagnosis
It will be necessary to carry out the differential diagnosis with acute mumps caused by other viruses, which present manifestations that can be superimposed on epidemic mumps. Suppurative parotitis, of bacterial etiology, produces a unilateral parotid swelling, associating a hot, erythematous and very painful swelling, together with high fever. Furthermore, in these cases, massage of the parotid gland makes it possible to extract purulent fluid through Stenon’s duct. Acute laterocervical adenitis can mimic mumps, which can be differentiated clinically by the location and absence of obliteration of the mandibular angle. Parotid gland enlargement, caused by drugs or metabolic disorders, is usually bilateral and asymptomatic. Other causes to consider in the differential diagnosis such as: tumors, cysts and obstruction caused by stones or stenosis, are usually unilateral.
Diagnosis
The characteristics described allow the diagnosis of suspected mumps to be established. In patients with typical symptoms, microbiological confirmation is not essential, but determination of the viral etiology helps define the current epidemiology of the infection. The confirmation of all sporadic cases is recommended. In outbreak situations, it is important to confirm most primary cases, unrelated to other cases, but it is not essential to confirm secondary cases.
Microbiological diagnostic tests are:
• Isolation of the virus in culture or amplification of nucleic acids, by reverse transcriptase polymerase chain reaction (RT-PCR, more sensitive than viral cultures) in: saliva samples, buccal mucosa (exudate from Stenon´s duct), CSF and urine, preferably in the first 3 days of the disease (since it loses a lot of sensitivity later).
• Serological study: determination of specific antibodies in the serum. The recommended sample for isolation and detection of mumps virus RNA is saliva.
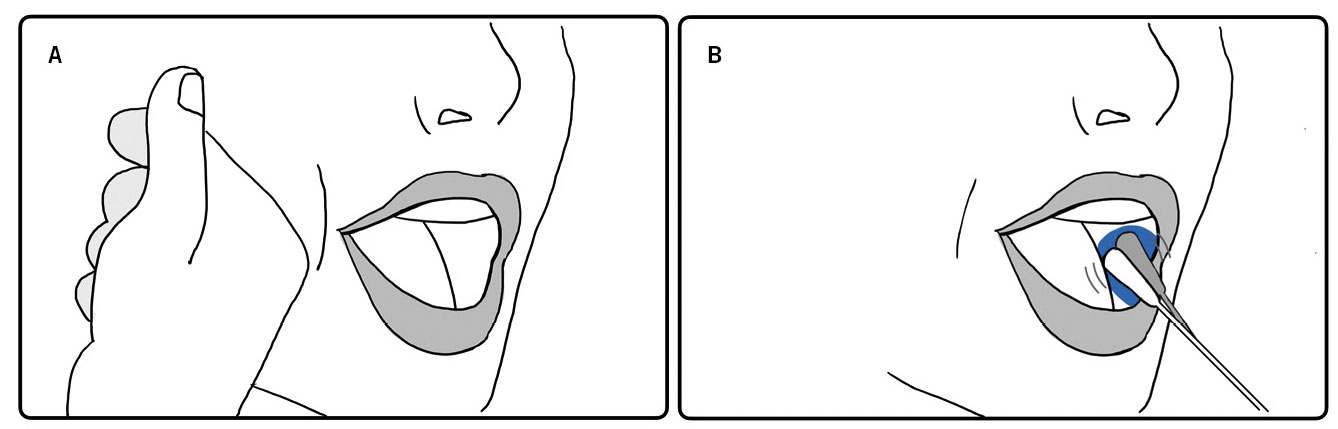
The saliva sample should be taken by massaging both cheeks, at the level of the parotid glands, for thirty seconds (Fig. 2A). Subsequently, rub the space of the oral mucosa between the cheek and the teeth, around Stenon’s duct, on both sides of the oral cavity with a swab (Fig. 2B).
Figure 2. Oral exudate of saliva collection technique in the study of mumps.
The swab will be immersed in virus transport medium and sent to the laboratory within 48 hours. In this video (https://youtu.be/ThvoJBjsUvQ) the sample extraction technique can be seen. If the initial IgM result is negative, a second sample obtained after 2-3 weeks should be tested, as some people, especially those who have been vaccinated, have none or late IgM response.
Other complementary tests can help guide the diagnosis. Ultrasound is a simple and affordable test that can confirm the presence of mumps in doubtful cases, as well as rule out other causes of parotid inflammation, such as sialolithiasis. Blood tests can also provide information. The white blood cell count and formula are usually normal, or there may be a mild leukopenia with relative lymphocytosis. Serum amylase levels are usually elevated for 2-3 weeks.
Treatment
Treatment of mumps is symptomatic and supportive. There is no specific treatment available for mumps virus infection. Analgesics relieve pain caused by inflammation of the salivary gland. Topical application of hot or cold compresses to the parotid gland can also relieve discomfort. It is advisable to avoid citrus foods. The treatment of orchitis is purely symptomatic, by means of: bed rest, analgesics, use of suspensory underwear for the testicles and the local application of cold compresses.
Cases with neurological symptoms usually require hospital admission for additional tests and surveillance.
Prevention
The most effective preventive measure is systematic vaccination(5). Vaccines against mumps are made up of an attenuated strain of this virus, combined with other components in the form of triple viral (measles, rubella and mumps) or tetraviral (also associated with varicella).
All mumps vaccines, with the exception of the one made with the Rubini strain, show a seroconversion rate of around 95% at 12 months after one dose. However, because some studies have shown lower efficacy, around 80% after a first dose, two-dose schedules are currently recommended.
The triple viral vaccine is indicated in all children from 12 months of age and in adolescents and adults who have been incorrectly vaccinated. The AEP (Spanish Pediatrics Association) Vaccine Advisory Committee and the Interterritorial Council of the National Health System recommend administering the first dose at 12 months and the second at 3-4 years of age. As with other live virus vaccines, triple viral and tetraviral vaccines are contraindicated in: pregnant women, patients undergoing immunosuppressive treatment, advanced malignant neoplasms or primary immunodeficiencies.
Vaccination and passive immunoprophylaxis after exposure have not been shown to reduce the risk of infection and are therefore not recommended. In case of outbreaks, the administration of a third dose can be considered, as recommended by the CDC (Centers for Disease Control and Prevention) in several recent outbreaks in university centers. Additionally, the temporary interruption of school attendance for all those children at risk due to incomplete immunization can be assessed.
Droplet isolation measures are indicated, along with standard precautions, in hospitalized patients with mumps, up to 5 days from the onset of the parotid mass. In the same way, school exclusion is recommended during the same period.
Other acute viral mumps
Other viruses have been implicated as causal agents of acute parotitis with negative results of the mumps virus tests, such as: parainfluenza virus, Epstein-Barr virus, influenza virus, rhinovirus or adenovirus(6). Due to systematic vaccination against mumps, these viruses have become more relevant. These infections present clinical characteristics similar to epidemic mumps.
Purulent sialadenitis
Unlike acute viral sialadenitis, purulent sialadenitis, caused by bacterial infections, produces a rapidly developing inflammation of the parotid gland, generally unilateral, associated with significant inflammatory signs of the skin, fever and, frequently, purulent suppuration through Stenon´s duct.
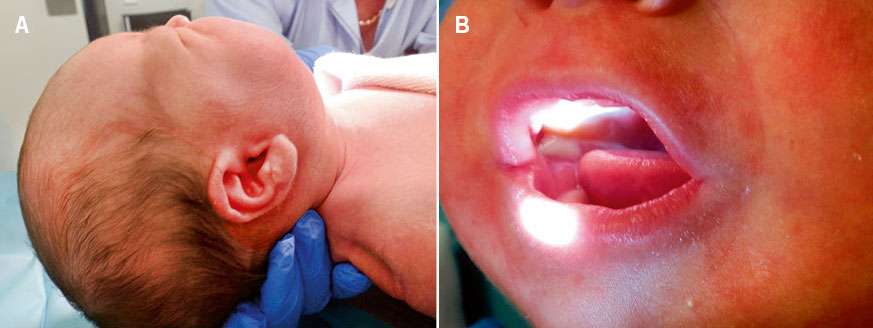
Also called suppurative parotitis, it is a bacterial infection of the salivary glands, more frequently of the parotid gland, which occurs acutely with painful swelling of the affected gland, associating: edema, erythema and heat in the skin, together with fever and malaise (Fig. 3A)(7). It is most often unilateral, but can also be bilateral.
The unilateral involvement and the rapid and obvious progress can help differentiate it from viral parotitis. On physical examination, the presence of inflammatory signs at the exit of Stenon’s duct is common. In addition, the outflow of purulent content through it when massaging the parotid gland is characteristic (Fig. 3B).
Figure 3. Neonatal suppurative parotitis due to S. aureus. A. Increase in size and inflammatory signs at the level of the mandibular angle. B. Purulent suppuration through Stenon’s duct. Image courtesy of Javier Nogueira López.
Infection can occur due to bacterial ascent through the salivary ducts from the oral cavity or via the hematogenous route. Conditions that produce thicker saliva or reduce saliva flow (e.g., dehydration, obstruction of the salivary ducts, autoimmune diseases such as Sjögren’s syndrome, drugs that cause xerostomia, etc.) facilitate its development. Inadequate oral hygiene, immunosuppression, diabetes mellitus, hypothyroidism, renal failure and the postoperative period have also been associated with a higher risk of presenting this disease.
The most frequently associated bacteria is Staphylococcus aureus, followed by anaerobic bacteria from the oral cavity, such as: Fusobacterium spp. and Peptostreptococcus spp. Different species of streptococci and gram-negative bacilli have also been implicated less frequently. Analytically, it generally highlights leukocytosis with a predominance of neutrophils. Ultrasonography allows confirmation of the diagnosis, ruling out predisposing anatomical factors (e.g., sialolithiasis) or intraparotid complications (e.g., parotid abscess). Although complications are unusual, in addition to abscess, the following have been described: osteomyelitis, thrombophlebitis of the jugular vein and sepsis.
The culture of the purulent exudate, obtained through the Stenon duct, allows us to guide the etiology, although it is often difficult to discern the role of some isolates, such as microorganisms that are usually commensals of the mouth, as it is a sample that can be contaminated during its extraction.
Treatment is mainly medical, with systemic antibiotic treatment, initially intravenous, ensuring good coverage against S. aureus (considering the possibility of methicillin-resistant S. aureus if there are risk factors) and anaerobes, such as amoxicillin-clavulanic acid. Some authors propose empirical coverage only against S. aureus (e.g., cloxacillin or cefazolin). The usual duration is 10-14 days. In addition, it is recommended to associate sialagogues (such as citrus juices), analgesia and, if possible, suspend the drugs that cause xerostomia. In case of parotid abscess, surgical drainage is indicated.
A specifically defined entity, although infrequent, is neonatal bacterial sialadenitis(8). It shares similarities in terms of etiological agents and clinical presentation with what is described above, but it is more frequently associated with complications such as sepsis. In addition to dehydration, other risk factors have been linked, such as prematurity or nasogastric feeding.
Granulomatous infections of the salivary glands
Children may infrequently be affected by granulomatous infections of the salivary glands, such as tuberculosis, cat scratch disease, actinomycosis, toxoplasmosis, or non-tuberculous mycobacterial infections(2). They usually have a course: subacute or chronic, indolent, slowly progressive and without significant inflammatory signs of the adjacent skin. Therefore, the differential diagnosis frequently includes neck tumors. Diagnosis of these diseases generally requires a biopsy.
Tuberculosis with extrapulmonary involvement of the salivary glands is very rare. It is more common in adolescents and adults. Occasionally, constitutional signs may be associated (fever, night sweats, and weight loss). The diagnostic and therapeutic approach is similar to pulmonary tuberculosis, with the particularity that the sample with the highest diagnostic yield will be the biopsy of the affected gland.
Sialadenitis due to non-tuberculous mycobacteria mainly affects young children, between 2 and 5 years of age. The violaceous color that the adjacent skin acquires is characteristic. Unlike tuberculous sialadenitis, the tuberculin skin test and interferon gamma release-based tests (IGRAs) are usually negative. There are different therapeutic approaches, from long-term medical treatment with different antibiotic regimens (macrolides, rifampicin, fluoroquinolones, ethambutol…), to complete removal of the gland.
Actinomycosis with involvement of the salivary glands, mainly the parotid gland, is rare. It is associated with poor dental hygiene. It produces a painless tumor, with a subacute course, without systemic symptoms. Frequently, it gives rise to skin fistulas. The presence of “sulfur granules” along with branching, filamentous, gram-positive rods at the periphery of the abscess pus is diagnostic. Treatment consists of prolonged antibiotic therapy with penicillin and/or amoxicillin, which includes a 2-6 weeks intravenous course, followed by oral treatment for 6-12 months. In case of poor response to antibiotic treatment, drainage or surgical debridement is required.
Toxoplasmosis occasionally produces intraparotid or periparotid lymphadenopathy, leading to an enlarged parotid gland. Cat scratch disease, caused by Bartonella henselae, can also infrequently affect the parotid gland.
HIV mumps
Among the different complications associated with HIV that can affect the salivary glands (e.g. Kaposi’s sarcoma, mycobacterial infections…), there is HIV-associated salivary gland disease, which consists of an increase in the size of these glands that can appear in the different phases of the infection, being, sometimes, the first presentation of the same. The most affected gland is the parotid gland, and the affectation is generally bilateral. It occurs due to the development of benign lymphoepithelial cysts within the gland.
Juvenile recurrent parotitis
Benign pathology in which recurrent episodes of mumps, of unknown cause, take place, generally improving with symptomatic treatment and tend to resolve spontaneously in adolescence.
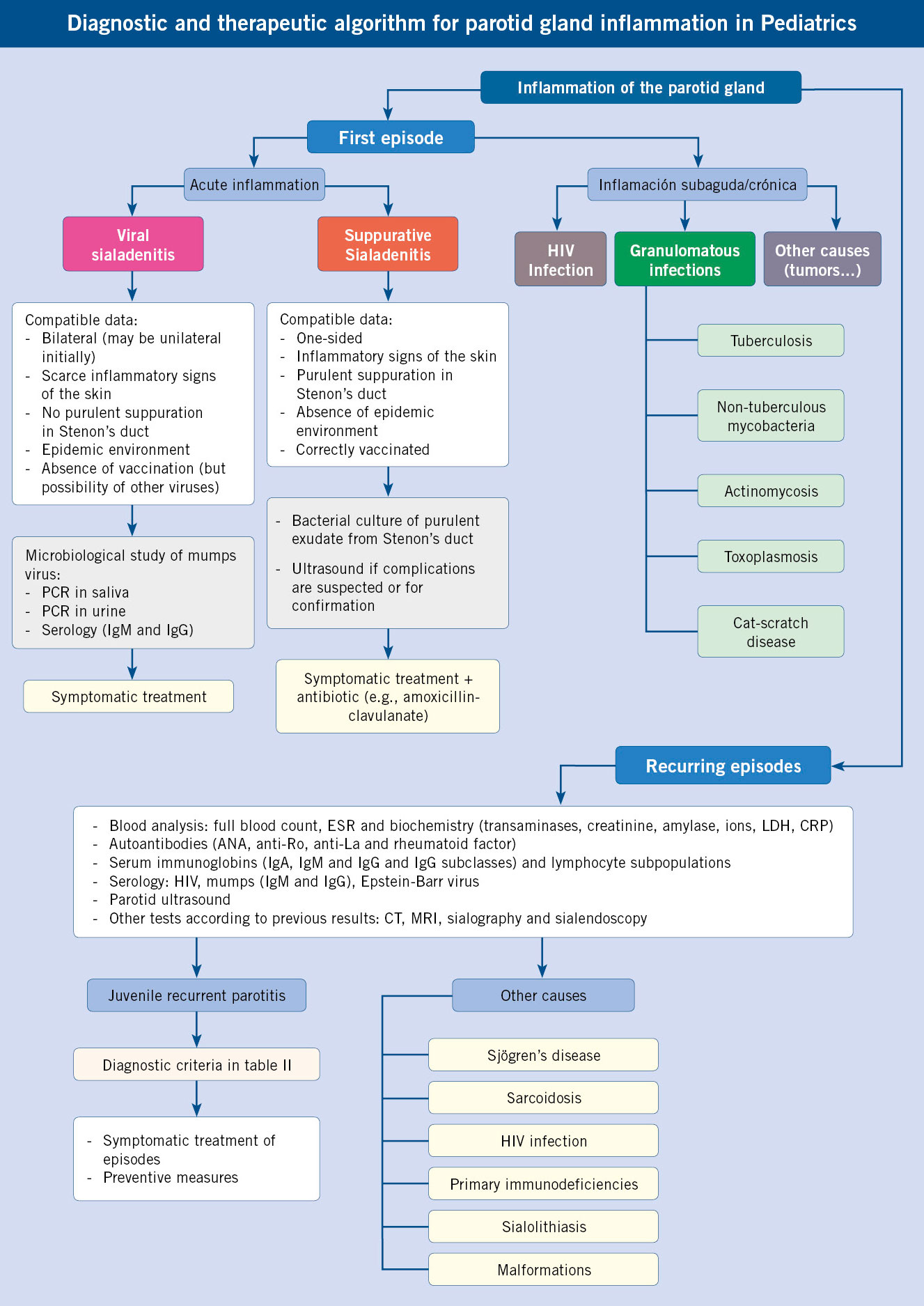
It is the second most common disease of the salivary glands. It is characterized by recurrent episodes of parotid inflammation, sometimes accompanied by fever, generally not high, and malaise(9). It is usually unilateral, presenting a more affected parotid gland when it is bilateral. It has a predominance in males. The age of onset shows a biphasic distribution, with an initial predominance between 2 and 6 years and, later, at puberty. Episodes generally last 4-7 days, with episodes typically occurring every 3-4 months. It frequently resolves in adolescence, although in some cases it extends into adulthood, with progressive deterioration of the glandular parenchyma. The diagnosis is clinical, excluding other possible causes: immunodeficiencies (mainly IgA deficiency, although it has also been associated with IgG3 deficiency), autoimmune diseases (Sjögren’s syndrome and sarcoidosis), infections (HIV) or obstructive alterations of the salivary ducts. Recently, some diagnostic criteria have been proposed (Table II).
The exact cause of this disease is unknown. Genetic factors have been formulated, as familial cases have been observed suggesting autosomal dominant inheritance with incomplete penetrance. In some cases, it has been related to anatomical factors, such as congenital anomalies of the salivary ducts. Also, a decrease in salivary flow that favors ductal dilation has been shown.
The role of bacteria is unclear. One study showed a high prevalence of bacterial isolates in oral samples in children with recurrent juvenile parotitis, but also in the control group of healthy children, which could correspond to normal colonization of the oral cavity.
Histologically, structural changes occur in the gland in the form of intraductal cystic dilatations with periductal lymphocytic infiltration, forming the so-called sialectasias. Whether they are primary or a consequence of repeated episodes is unknown.
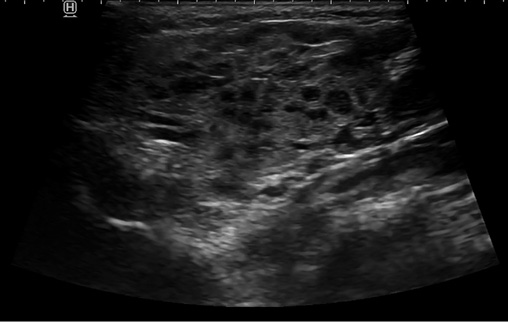
Ultrasound (Fig. 4) is a simple test that can identify sialectasias and rule out other causes, such as obstructive disorders (e.g., sialolithiasis), tumor lesions, or vascular malformations. Other imaging tests, such as CT, nuclear magnetic resonance or sialography, can be assessed in certain cases (suspicion of complications, definition of lesions observed on ultrasound or as an additional therapeutic approach in the case of sialography).
Figure 4. Multiple hypoechoic images corresponding to sialectasias in a child with recurrent juvenile parotitis.
The anamnesis must include: the history of recurrent infections and/or atypical course, which could lead to a primary immunodeficiency; risk factors for HIV infection, both postnatal acquisition (risky sexual practices, transfusions of blood products without health control) and vertical acquisition (maternal HIV infection, pregnancy follow-up); and the presence of a family history of autoimmune diseases and other systemic symptoms, which could point to an autoimmune disease.
The initial evaluation should include: autoantibody study: antinuclear (ANA), anti-Ro (SS-A), anti-La (SS-B) and rheumatoid factor; and a study of immunodeficiencies (quantification of serum immunoglobulins, including IgG subclasses and lymphocyte subpopulations).
During episodes, analgesics and anti-inflammatory drugs are used to alleviate symptoms(10). Massage and local heat on the parotid gland can relieve discomfort. The use of sialogogues, such as citrus juices, is proposed to increase the flow of saliva, but can sometimes exacerbate the pain. Some authors have proposed the use of short regimens of systemic corticosteroids in the acute phase, after having previously ruled out other diagnoses (e.g., lymphoma). The role of antibiotics is controversial. However, in case of suspected bacterial superinfection (purulent suppuration through Stenon’s duct, persistent high fever, significant elevation of acute phase analytical reactants, parotid abscess formation, clinical deterioration), its use is recommended, following the same indications as in acute purulent sialoadenitis.
To try to avoid new episodes, maintaining a good oral hygiene and adequate hydration is recommended. In some cases of very frequent episodes that seriously affect the patient, sialendoscopy with probing, lavage and dilation of the duct may be indicated. In patients with persistent symptoms or if episodes continue into adulthood, the following may be considered: ductal ligation, tympanic neurectomy, or even total parotidectomy, although potential risks, such as facial nerve involvement, should be taken into account.
Non-infectious inflammatory diseases of the salivary glands
Although rare in children, several diseases, such as Sjögren’s syndrome or sarcoidosis, can affect the salivary glands. They must be taken into account in the differential diagnosis of chronic or recurrent sialadenitis.
Sjögren’s syndrome
Chronic autoimmune disorder of the exocrine glands that can present systemically, with involvement of multiple glands, or locally, with exclusive involvement of the salivary and lacrimal glands. It usually presents in adulthood, but occasionally symptoms begin around 10 years of age. The disease is characterized by lymphoplasmacytic infiltration with resulting glandular hypofunction, leading to dry mouth and eyes. Its etiology is multifactorial and involves an interaction between genetics, the immune system, and the patient’s environmental exposures.
The predominant clinical presentation is dryness of the mouth and eyes. Xerostomia results in: difficulty chewing and swallowing food, difficulty with phonation, dental caries and adherence of food to the oral mucosa. Chronic irritation and destruction of the corneal and conjunctival epithelium cause keratoconjunctivitis sicca. Enlargement of the salivary glands, most commonly the parotid glands, occurs in 25-66% of patients. This may start unilaterally, but most patients eventually develop bilateral enlargement, which may be recurrent or chronic.
Systemic manifestations include: malaise, fever, myalgias and arthralgias. Some organs affected in systemic forms are: kidneys (tubular acidosis), skin (Raynaud’s phenomenon due to vasculitis) and central nervous system (peripheral sensory and motor polyneuropathies). Sjögren’s syndrome also predisposes to the development of malignant lymphoproliferative disorders.
Diagnosis includes the following findings: keratoconjunctivitis sicca; decreased salivary and tear flow; characteristic histologic findings on biopsy of the minor salivary gland; and detection of anti-Ro (SS-A) and anti-La (SS-B) serum autoantibodies. Dry eyes and mouth take years to appear when they start in childhood, so diagnostic criteria are less useful in childhood. The treatment is both symptomatic and preventive of irreversible damage to the different organs, mainly the teeth and eyes.
Sarcoidosis
Systemic disease that causes the formation of granulomas in various organs, especially in the lungs and lymphatic organs. In 5-10% of cases, the salivary glands are affected, causing their inflammation. One particular presentation, uveoparotid fever or Heerfordt’s syndrome, produces chronic inflammation of the parotid glands along with uveitis and facial nerve paralysis.
Other inflammatory diseases
Although more infrequent, other diseases such as granulomatosis with polyangiitis, Rosai-Dorfman disease and Kimura disease can affect the salivary glands, leading to their inflammation and enlargement.
Obstructive pathology of the salivary glands
Ranulas and mucoceles are a consequence of the obstruction of the submandibular/sublingual glands and the minor glands, respectively, and are typical of children, unlike lithiasis, which are very infrequent. Sialendoscopy allows the diagnosis and treatment of various glandular pathologies.
Cystic pathology: ranulas and mucoceles
Ranulas (Fig. 5) are pseudocysts due to mucous or salivary retention, secondary to alterations of the excretory ducts (post-traumatic or obstructive) in the sublingual or submaxillary glands(11).
Figure 5. Sublingual ranula in an 11-year-old girl. Image courtesy of Alicia Martínez Sebastián.
In children, the most common are simple ranulas restricted to the floor of the mouth. Those that extend to the cervical/parapharyngeal spaces are rare at this age. The highest incidence occurs in the first and second decades of life. Clinically, ranulas manifest as: inflammation of the gland whilst chewing or in situations of stimulation of saliva production(12). They can cause induration of the floor of the mouth and the appearance of cysts with mucous/salivary content of greater or lesser size.
When the obstruction affects a minor salivary gland, they are called mucoceles, and the cause is the same. Mucoceles appear as cystic lesions in any location of the oral cavity, although they are also common on the lips.
The treatment of ranulas can range from puncture and aspiration, marsupialization and infiltration of sclerosing substances, to excision of the complete sublingual gland(13). Mucoceles are usually monitored and, depending on the clinical picture, the entire gland can be drained or resected.
Glands with cervical extension are more complicated to manage and require imaging tests (CT or MRI) to make a differential diagnosis with other cystic lesions, such as lymphatic malformations. The age of presentation, as well as the course of the disease, will guide us on the diagnosis. Rapidly evolving lesions tend to be of infectious/inflammatory origin. Regarding age, tumors in children under one year of age tend to have a vascular origin, while in older children they are usually solid tumors.
Sialolithiasis
It is very rare in children, less than 3% of all cases are in pediatric patients. It is much more common in submandibular glands than in parotid glands(14). It must be suspected in the event of recurrent episodes of unilateral glandular inflammation (therefore a differential diagnosis with recurrent parotitis must be performed).
Occasionally, the stone can be seen at the outlet of Wharton’s duct, in the floor of the mouth, but if it is more distal, imaging tests are necessary. The classic treatment has been marsupialization of the ducts to extract the stones and even complete excision of the glands.
In recent years, sialoendoscopy has been developed as a diagnostic and therapeutic technique for salivary gland pathology, with a progressive increase in its use in children(15). It consists of the dilation of the papillae with specific dilators and the subsequent introduction and use of an endoscope designed for this purpose, to explore the submandibular and parotid glands, accessing through the Wharton and Stenon ducts, respectively, allowing navigation through the branches of the ducts (diagnostic application) and carry out treatments such as: application of intraductal medication (corticosteroids and/or antibiotics), washing, dilation of stenosis and removal of stones. It is a safe technique that is almost always performed under general anesthesia and with good results and few complications.
The most frequent indication for sialendoscopy is recurrent juvenile parotitis, followed by lithiasis and, much less frequently, by cases of stenosis.
Tumor pathology
Benign tumors
Parotid hemangioma is the most frequent glandular tumor in Pediatrics. With regard to solid tumors, the most frequent is the pleomorphic adenoma, a benign tumor, but with a tendency to recur and malignant potential.
They arise mainly in the parotid, followed by those that affect the submandibular glands, minor and sublingual glands(16). The most frequent type of solid tumor is the pleomorphic adenoma and, globally, the hemangioma is the most frequent of all parotid tumors in childhood.
Epithelial tumors: pleomorphic adenoma
It is the most frequent tumor of the salivary glands. It is benign, but has a series of characteristics that condition its management:
• Recurrence tendency.
• Possible involvement of the facial nerve in its treatment.
• Malignancy potential
Depending on its anatomical origin, its form of presentation will change. In the parotid or submandibular gland, they will present as: single lesions, with progressive growth, of firm consistency, but mobile and of variable size according to progress. When they originate in a smaller gland, they are usually smaller in size, hard and sticky in consistency. They are painless and do not tend to have skin inflammation.
Contrast-enhanced MRI is the imaging technique of choice. Characteristically, it has a hypointense peripheral ring that helps in its diagnosis, although it is not always present. Ultrasound has more limited utility, although it is more accessible and easier to perform without sedation in younger children. It is common to perform ultrasound first and then MRI to obtain more precise information, in necessary cases. Histological diagnosis by fine-needle aspiration (FNA) helps in decision-making.
The treatment of choice is surgery with wide resection margins. Despite being a benign tumor, the potential for malignancy and the possibility of recurrence require an excision with safety margins, this being the minimum acceptable surgery. The problem with pleomorphic adenoma surgery is given by the possible sequelae of treatment despite being a benign tumor, such as neurovascular lesions or aesthetic defects.
Benign mesenchymal tumors
Parotid hemangioma is the most common tumor of the parotid gland in childhood. They account for 90% of salivary tumors in children under one year of age and are usually associated with other hemangiomas in the head and neck.
They are soft, non-compressible lesions that generate changes in the overlying skin, so that it acquires a purplish-blue hue. The consistency can be variable; in some areas it can be softer and in others harder if there are fibrosis phenomena.
The size varies from one patient to another, but it has the potential to grow and cause aesthetic and/or functional involvement depending on the affected structures, although it does not usually cause facial paralysis.
The first diagnostic technique that is usually used is ultrasound, which shows hypoechoic lesions with respect to healthy glandular tissue, but the most useful study is MRI, which will provide a lot of information regarding extension, location, etc.
Management will depend on the size and progress; since, in general, they tend to regress, so in small lesions and/or without growth, a wait-and-see attitude can be maintained, while in large tumors, with rapid growth or that produce symptoms, the treatment of choice is propranolol, with which a response has been shown in almost 90% of children in the first 5 weeks of treatment. Surgery is reserved for cases in which medical treatment fails.
Lymphatic malformations may originate primarily in the glands (particularly parotid) or be affected by large lesions originating in other locations. They can give rise to important aesthetic and/or functional alterations depending on their extension. Treatment is usually through sclerotherapy and/or surgery depending on the location and extension. They can give rise to phenomena of inflammation with rapid increases in size.
Malignant tumors
They are exceptional in Pediatrics and tend to preferentially affect the parotid gland. In addition, the probability that a parotid mass is malignant is higher than in adults. The prognosis depends on histology. Treatment is surgical and radio-chemotherapy protocols are not well established due to the rarity of this pathology.
Generalities
The characteristics of these tumors are different in children and adults:
• In children, the percentage of malignant lesions over the total number of salivary gland tumors is higher than in adults (a tumor is more likely to be malignant than in adults).
• In children, malignant tumors affect the parotid more than the submandibular gland.
• Mesenchymal tumors are more common than in adults (lesions of vascular origin are the most frequent).
• In children, well-differentiated tumors (better prognosis) are more common than in adults.
Regarding histology, the most frequent malignant solid tumors are: mucoepidermoid carcinomas (40-92% of carcinomas), followed by acinar cell carcinomas, adenocarcinoma and adenoid cystic carcinoma. In the minor and sublingual glands, benign tumors are more frequent than malignant ones, among which mucoepidermoid carcinoma is also the most frequent(17).
The presence of distant metastases at diagnosis is rare, and cervical lymph node extension is infrequent. The development of these neoplasms has been related to infection by the Epstein-Barr virus and previous treatments with radiotherapy and/or chemotherapy, although the vast majority of cases are of unknown origin.
Clinical presentation
They are single masses of slow and progressive growth, generally painless and without inflammatory signs(18). The presence of pain is characteristic of adenoid cystic carcinoma. Clinical presentation with facial paralysis or the presence of cervical lymphadenopathy is very infrequent. In the case of minor salivary glands, they appear more frequently in the oral cavity or as a tumor on the floor of the mouth in the case of sublingual glands.
More than 90% of patients are over 10 years of age at the time of diagnosis, with no clear gender predominance. When they appear in children under 10 years of age, they tend to be higher-grade tumors with a worse prognosis.
Diagnosis
The presence of a persistent, progressively growing tumor should make us suspect a possible neoplasm(19). The imaging technique of choice is MRI with contrast.
The initial histological diagnosis is usually made with FNA, which will allow differentiation between a malignant tumor and other types of tumors (abscess, lymphoma, hemangioma, lymphatic malformation). However, it will not be useful to distinguish between different types of malignant tumors (it only agrees 40% with the definitive diagnosis of the surgical piece).
The performance of open biopsies of the lesions is not usually indicated, unless the size of the lesion prevents a complete resection and in order to propose alternative treatments to surgery.
Treatment
Surgical resection with wide margins is the therapeutic mainstay for these tumors. The overall prognosis of these tumors is good(20).
Role of the Primary Care pediatrician
Most of the children with acute involvement of the salivary glands, treated in Primary Care, correspond to cases of viral parotitis, in some cases epidemic parotitis. It is a notifiable disease, so its communication and the correct collection of samples, usually with the support of Public Health, can help its epidemiological surveillance. In addition, preventive measures are essential to avoid the transmission of this infection.
Juvenile recurrent parotitis is a disease that, despite its benignity, produces significant anguish in the family and the patient. For this reason, it is essential to know how to explain its characteristics and carry out a correct follow-up.
On the other hand, in the differential diagnosis of these pathologies, we must not forget other diseases, such as Sjögren’s syndrome or tumor diseases that affect the salivary glands, which will require their referral for evaluation by a specialist.
Conflict of interests
There is no conflict of interest in the preparation of the manuscript.
Bibliography
Asterisks reflect the interest of the article in the opinion of the authors.
1.*** Jackson NM, Mitchell JL, Walvekar RR. Inflammatory Disorders of the Salivary Glands. En: Flint P, Haughey B, Valerie L, et al. Cummings Otolaryngology Head and Neck Surgery. Seventh Edition. Elsevier; 2020. p. 1157-70.
2. Viñallonga Sardá X. Patología de las glándulas salivales (Pathology of the salivary glands). Pediatr Integral. 2017; XXI: 474-83.
3.** Litman N, Baum SG. Mumps virus. In: Bennett JE, Dolin R, Martin J. Mandell, Douglas, and Bennett. Infectious diseases. Principles and practice. 9th edition. Elsevier; 2020. p. 2087-92.
4.** Cherry JD, Quinn KK. En: Mumps Virus. Feign. Cherry J, Demmler-Harrison GJ, Kaplan SL, Steinbach WJ, Hotez P. Feigin and Cherry’s Textbook of Pediatric Infectious Diseases. Eighth Edition. Elsevier; 2019. p. 1771-9.
5. Vaccine Advisory Committee (CAV-AEP). Parotitis. AEP Online Vaccine Manual (Internet). Madrid: AEP; 2022. Accessed March 1, 2022. Available at: http://vacunasaep.org/documentos/manual/cap-32.
6. Elbadawi LI, Talley P, Rolfes MA, Millman AJ, Reisdorf E, Kramer NA, et al. Non-mumps Viral Parotitis During the 2014-2015 Influenza Season in the United States. Clin Infect Dis. 2018; 67: 493-501. DOI: 10.1093/cid/ciy137.
7. Shaw J. Infections of the Oral Cavity. En: Long SS, Prober CG y Fischer M. Principles and Practice of Pediatric Infectious Diseases. Fifth Edition. Elsevier; 2018. p. 93-199.e2.
8. Martínez de Morentin Navarcorena AL, Nogueira López J, Gutiérrez Martín A. Fiebre en el neonato. Si no lo veo, no lo creo. En: Imagen de la semana (Fever in the newborn. If I do not see it, I do not believe it). In: Image of the week. Continuum 2022 (online). Retrieved May 14, 2022. Available at: http://continuum.aeped.es.
9.*** Wood J, Toll EC, Hall F, Mahadevan M. Juvenile recurrent parotitis: Review and proposed management algorithm. Int J Pediatr Otorhinolaryngol. 2021; 142: 110617. DOI: 10.1016/j.ijporl.2021.110617.
10. Garavello W, Redaelli M, Galluzzi F, Pignataro L. Juvenile recurrent parotitis: A systematic review of treatment studies. Int J Pediatr Otorhinolaryngol. 2018; 112: 151-7. DOI: 10.1016/j.ijporl.2018.07.002.
11. Bachesk AB, Bin LR, Iwaki IV, Iwaki Filho L. Ranula in children: Retrospective study of 25 years and literature review of the plunging variable. Int J Pediatr Otorhinolaryngol. 2021; 148: 110810. DOI: 10.1016/j.ijporl.2021.110810.
12. Martínez Sebastián A, Martínez Sebastián L. Tumoración sublingual en adolescente de rápido crecimiento (Rapidly growing sublingual tumor in an adolescent.). Pediatr Integral. 2021; XXV: 443.e1-e6.
13.** Zhi K, Gao L, Ren W. What is new in management of pediatric ranula? Curr Opin Otolaryngol Head Neck Surg. 2014; 22: 525-9. DOI: 10.1097/MOO.0000000000000103.
14. Nahlieli O, Eliav E, Hasson O, Zagury A, Baruchin AM. Pediatric sialolithiasis. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2000; 90: 709-12. DOI: 10.1067/moe.2000.109075a.
15. Schwarz Y, Bezdjian A, Daniel SJ. Sialendoscopy in treating pediatric salivary gland disorders: a systematic review. Eur Arch Otorhinolaryngol. 2018; 275: 347-56. DOI: 10.1007/s00405-017-4830-2.
16.** Bradley PJ, Eisele DW. Salivary Gland Neoplasms in Children and Adolescents. Adv Otorhinolaryngol. 2016; 78: 175-81. DOI: 10.1159/000442138.
17.** Ord RA, Carlson ER. Pediatric Salivary Gland Malignancies. Oral Maxillofac Surg Clin North Am. 2016; 28: 83-9. DOI: 10.1016/j.coms.2015.07.007.
18. Yoshida EJ, García J, Eisele DW, Chen AM. Salivary gland malignancies in children. Int J Pediatr Otorhinolaryngol. 2014; 78: 174-8. DOI: 10.1016/j.ijporl.2013.11.001.
19. Rebours C, Couloigner V, Galmiche L, Casiraghi O, Badoual C, Boudjemaa S, et al. Pediatric salivary gland carcinomas: Diagnostic and therapeutic management. Laryngoscope. 2017; 127: 140-7. DOI: 10.1002/lary.26204.
20. Radomski S, Dermody S, Harley EH Jr. Clinical characteristics and outcomes of major salivary gland malignancies in children. Laryngoscope. 2018; 128: 1126-32. DOI: 10.1002/lary.26946.
Recommended bibliography
– Chafin JB, Bayazid L. Pediatric Salivary Gland Disease. Pediatr Clin North Am. 2022; 69: 363-80. DOI: 10.1016/j.pcl.2022.01.004.
Updated review that includes the main pathologies of the salivary glands in the pediatric population.
– Mumps surveillance protocol. In: National Center for Epidemiology. Carlos III Health Institute. National Epid Surveillance Networkemiological. Protocols of the National Epidemiological Surveillance Network. Madrid; 2013 (reviewed in September 2016). p. 444-52. Available at: https://www.isciii.es/QueHacemos/Servicios/VigilanciaSaludPublicaRENAVE/EnfermedadesTransmisibles/Documents/PROTOCOLOS/PROTOCOLOS%20EN%20BLOQUE/PROTOCOLOS_RENAVE-ciber.pdf.
Epidemiological surveillance protocol of the Spanish National Center for Epidemiology, which details the diagnostic criteria and the microbiological tests to be performed in cases with a suspected or confirmed diagnosis of epidemic parotitis.
| Clinical case |
|
A 7-year-old male, with no relevant personal history, correctly vaccinated, attends the Primary Care consultation due to a bilateral painful tumor in the mandibular angle of 24 h of evolution, associated with low-grade fever and rhinorrhea. On examination, he presents a tumor at the level of both parotid glands and erythematous edema at the level of the orifice of Stenon’s duct, without suppuration through it, and with a few inflammatory signs in the adjacent skin. There is no epidemic environment.
|
 Salivary gland pathology
Salivary gland pathology