|
| Topics on Continuous Training |
J.M. Azaña Defez, M.L. Martínez Martínez
Doctors in Medicine and Surgery. Consultant Physicians at the Dermatology Service. Pediatric Dermatology Unit. University Hospital Complex of Albacete
| Abstract
Acne is a chronic inflammatory skin disease of the pilosebaceous unit of multifactorial etiology characterized by increased sebaceous secretion, comedone formation, inflammatory lesions and risk of scarring sequelae. It is undoubtedly one of the most frequent dermatological processes in the daily clinical practice, especially in adolescence, although it can also appear in childhood and persist into adulthood. Adequate management of this pathology is relevant, as it can cause lower self-esteem and social dysfunction in patients, with the subsequent impact on quality of life. |
| Abstract
Acne is a chronic inflammatory skin disease of the pilosebaceous unit of multifactorial etiology characterized by increased sebaceous secretion, comedone formation, inflammatory lesions and risk of scarring sequelae. It is undoubtedly one of the most frequent dermatological processes in the daily clinical practice, especially in adolescence, although it can also appear in childhood and persist into adulthood. Adequate management of this pathology is relevant, as it can cause lower self-esteem and social dysfunction in patients, with the subsequent impact on quality of life. |
Key words: Cutibacterium acnes; Grading and classification of acne; Acne management; Isotretinoin.
Palabras clave: Acné; Cutibacterium acnes; Graduación y clasificación del acné; Manejo del acné; Isotretinoina.
Pediatr Integral 2021; XXV (4): 166 – 175
 |
 |
Acne
Introduction
Acne is a frequent inflammatory skin disease of chronic course and polymorphous in its clinical expression.
Acne is a chronic skin disease of the pilosebaceous unit, of multifactorial etiology, characterized by its clinical polymorphism. It undoubtedly represents one of the dermatological processes of greatest interest in daily clinical practice.
Epidemiology
Acne can appear in all stages of life, although its prevalence is higher in adolescence and there seems to be a genetic predisposition.
Acne is one of the most frequent dermatological diseases, as it is estimated that around 85% of the population will present it throughout their lives(1). The prevalence of acne among the Spanish population aged 12 to 18 years is 74%, without significant differences regarding sex and with a peak between 14 and 16 years of age(2); therefore, it accounts for 25% of dermatological consultations. Acne is estimated to be of moderate / severe intensity in about 20% of patients.
Its highest prevalence and intensity occur around 14-15 years of age in females and somewhat later (16-18 years) in males. Despite its prevalence in adolescence, in 7-25% of patients it will persist into adulthood.
In females, it can manifest a longer course; whilst in males, more serious forms are identified(3).
There seems to be certain genetic predisposition to develop acne: history of acne is often found in parents, and in addition, there is a high concordance in monozygotic twins(3,4).
Seasonal variations in acne severity are observed, with a tendency to worsen in winter(5).
Etiopathogenesis
Acne is a multifactorial disease, produced by: increased sebaceous secretion, follicular epidermal hyperproliferation, comedogenesis, bacterial colonization and induction of inflammation.
Acne is a disease of the pilosebaceous follicle, induced by androgens of adrenal and gonadal origin: its onset correlates with the increase in sebaceous production triggered by this hormonal stimulus. The pilosebaceous unit is the target organ of acne, explained by the distribution of lesions in the areas with the highest concentration.
The factors involved in its development include: increased sebaceous secretion and follicular epidermal hyperproliferation, which leads to the formation of: comedones (comedogenesis), bacterial colonization by Cutibacterium acnes (formerly named Propionibacterium acnes) and induction of inflammation(6).
The onset and persistence of the activity of the sebaceous glands is mainly due to the action of androgens. After its activity in the postnatal period, due to the maternal hormonal influence, sebaceous glands remain minimized until puberty, when the size and number of lobes per gland increases, as a result of the androgenic stimulus (adrenarche). The presence of this glandular activity is a necessary requirement for the development of acne. There is a greater sebaceous secretion and, in addition, a qualitative alteration. These events have been related to the development of hypercornification of the sebaceous duct and changes in surface microorganisms. The consequence of hypercornification of the sebaceous duct is microcomedone: keratinocytes are grouped in dense clumps with monofilaments and lipid droplets, retaining the secreted sebum that distends the channel and the gland. This microcomedone is the primary acne lesion.
Acne is not an infectious process, but there are microorganisms that colonize and multiply in the follicular duct, and that can play a role in the pathogenesis. Cutibacterium acnes (C. acnes), which predominates in areas rich in sebaceous glands, can act as an opportunistic pathogen in acne. C. acnes is scantily present on the skin surface, whereas it is the dominant resident in the pilosebaceous unit; and the development of acne would be related, not with its proliferation, but with the selection of certain types, especially the IA1 phylotype, in a medium with increased sebaceous secretion in addition to balance alteration of the skin microbiome(7,8). Also, the formation of biofilm, an organized conglomerate of bacteria enhancing their survival, could increase its pathogenicity and resistance to antibiotics. These data open up new therapeutic possibilities in the management of acne (probiotics, anti-biofilm compounds…)(9).
The inflammation would not be caused by the presence of bacteria in the dermis, but by the action of biologically active mediators produced by C. acnes, which diffuse from the follicular channel and, later, by an inflammatory reaction to a foreign body triggered by the rupture of the ductal wall. The cell wall of C. acnes contains a carbohydrate antigen that stimulates the development of antibodies, which facilitate the inflammatory response.
There are multiple factors related to acne exacerbation episodes. Various studies postulate that diet could be considered a stimulating factor for acne, since foods that are high in sugar and other carbohydrates, dairy products or proteins, would affect serum insulin and insulin-like growth factor (IGF-1), which would induce an increase in the production of available androgens and the development of acne(3). However, currently, there is no scientific evidence with controlled studies to justify the restriction of specific foods(10).
The exacerbation of lesions in stressful situations is a known fact, in relation to the neuroendocrine regulation of sebocytes, the increase in adrenal secretion and the manipulation of lesions. Conversely, acne has an undoubted psychosocial impact, with consequent repercussions on quality of life.
About 70% of patients report a premenstrual acne exacerbation. This has been related to: increased hydration of the pilosebaceous duct, progressive decrease in estrogen levels with anti-inflammatory action and increased progesterone in this phase, with androgenic and pro-inflammatory effects.
70% of patients show acne improvement with sun exposure during the summer months. This amelioration could be potentially explained by the “camouflage” effect caused by tanning and an anti-inflammatory action of the immune suppression induced by ultraviolet light(5). This is the theoretical foundation of phototherapy for acne. However, ultraviolet radiation can also increase the comedogenic effect of sebaceous secretion and cause significant exacerbations of acne (“acne Mallorca”, acne aestivalis, tropical acne).
Activities in humid climates and occlusion determine acne worsening in up to 15% of patients. It could be due to a ductal hydration mechanism that would favor the obstruction. This is the mechanism that explains the exacerbation of acne lesions produced by the use of protective masks during the SARS-CoV-2 pandemic (“Mask acne”).
Various drugs, such as anabolic steroids or contraceptives containing progestogens with androgenic action, can exacerbate acne. In addition, there are numerous treatments such as: oral corticosteroids, isoniazid, lithium or certain anti-cancer drugs, capable of inducing acneiform eruptions.
The use of high lipid-based cosmetic formulations, aggressive cleaners or alkaline soaps can: alter the skin barrier, promote the formation of comedones and induce inflammation.
The term exposome defines the assorted environmental factors that influence the development and severity of a disease, in this case acne. Identifying the negative exposome factors can help reduce its impact and manage the disease(11).
Clinical manifestations
Acne is an inflammatory skin disease with two main characteristics: lesional polymorphism and chronic course.
In addition to seborrhea, open and/or closed comedones and inflammatory lesions are observed such as: papules, pustules and nodules, as well as residual lesions (scars and pigmentation alterations).
Regarding the distribution of the lesions, almost all the patients (99%) present facial localization, accompanied in more than half of the cases, by involvement of the back (60%) and, to a lesser extent, the pectoral area (15%). The majority of patients will refer to a gradual onset of the lesions around puberty, so that in cases in whom a sudden onset of these is described, an underlying cause must be ruled out. In women with severe, rapid-onset acne associated with hirsutism or menstrual irregularities, the existence of endocrinological pathology (hyperandrogenism) must be excluded.
Types of lesions
Non-inflammatory lesions
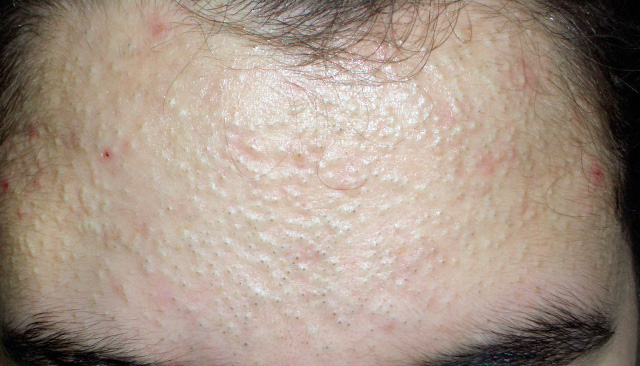
Closed comedones are small firm papules with a whitish surface in which, occasionally, the follicular opening is observed. Alternatively, open comedones present a blackish central plug, due to the deposit of melanin and the oxidation of sebum (Fig. 1).
Figure 1. Comedonian acne: open and closed comedones (“blackheads”).
Inflammatory lesions
They derive from the previous ones and include:
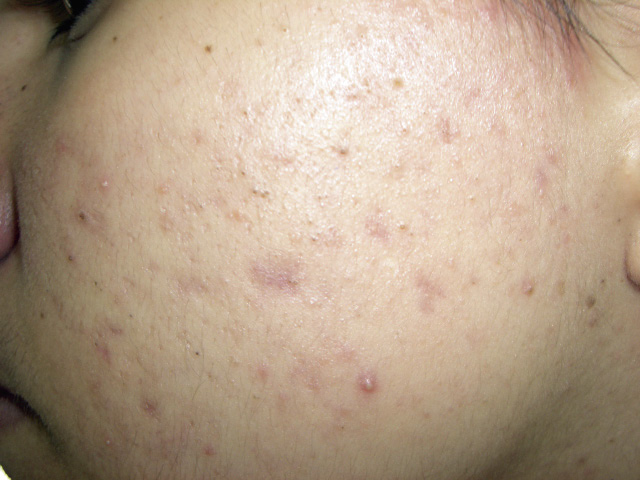
• Superficial lesions: papules and pustules (Fig. 2).
Figure 2. Mild-moderate inflammatory acne: comedones, papules and pustules.
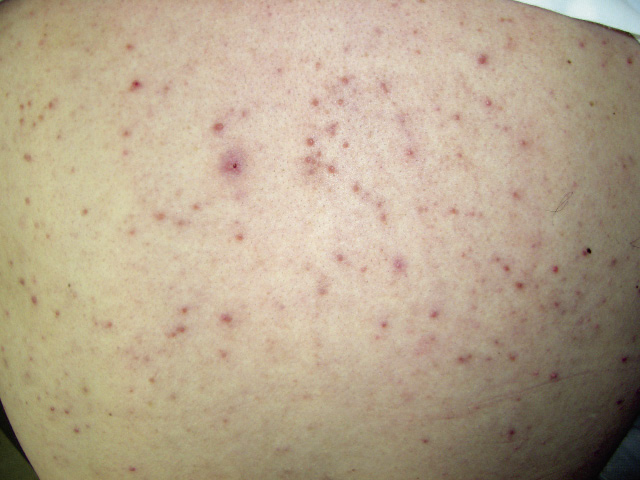
• Deep lesions: deep pustules and nodules, when the inflammation affects the entire follicle. Nodules are firm, painful lesions that can be larger than 1 cm. They constitute the characteristic lesion of what is known as acne conglobata. Although epidermoid cysts can be seen in patients with acne, in most cases, the so-called “cysts” are not true cyst, but rather deep nodules (nodular acne).
Residual injuries
Residual lesions are observed in up to 90% of patients, although they significant in only 22% of cases are. Inflammatory lesions can leave sequelae such as erythematous macules that can persist for months, scars of various characteristics (depressed, varioliform, ice pick, hypertrophic, keloid) and dyschromias (post-inflammatory hyperpigmented and hypopigmented macules).
Types of acne
Based on the clinical presentation and the predominance of some of these lesions, classic clinical types of acne are distinguished: comedonian, papule-pustular and nodular. Acne conglobata is a severe and treatment-resistant form, more frequently found in men and with a more intense location on the trunk. It is characterized by deep papules and painful nodules, which can converge and form sinusoidal paths, and evolve to form depressed and keloid scars. Acne fulminans or malignant acne, initially described as a form of acne conglobata (“acute febrile ulcerative acne conglobata”), is a rare variant in which a systemic immune reaction against C. acnes takes place(12). It is observed especially in young men who, suddenly, develop inflammatory lesions predominantly in the trunk, with systemic manifestations (fever, anorexia, polyarthropathy, splenomegaly, aseptic osteolysis), together with marked leukocytosis. Blood cultures are sterile. Sometimes it can be induced by drugs (isotretinoin or androgens) and intercurrent infections (Epstein-Barr virus). In terms of treatment, in addition to that indicated for acne, the use of oral corticosteroids is required.
Special forms of acne of interest in Pediatrics
Infantile acne
It has been associated with transplacental stimulation of adrenal androgen production, at least in cases of onset before the first year of life. There are no other manifestations of hyperandrogenism, because sebaceous glands are the only ones capable of converting dehydroepiandrosterone to androstenedione and testosterone. Occasionally, drugs such as phenytoin, corticosteroids and halogens have been implicated.
It prevails in males, usually as a localized form, with especial involvement of the cheeks. In the neonatal period, the main differential diagnosis must be established with benign cephalic pustulosis, which is much more frequent (20% of neonates), with onset within the first weeks of life, consisting of papulopustular lesions on the cheeks, but without comedones and associated with colonization by Malassezia (M. sympodialis and M. furfur).
The treatment of infantile acne is similar to that of acne vulgaris (see below), but excluding oral tetracyclines. Only exceptionally has oral isotretinoin been used in this form(13).
Excoriated acne
Although it is more frequent in young women, it can also be diagnosed in adolescence in patients who manipulate the lesions, causing erosions or ulcerations with the risk of superinfection and scarring. The most severe cases can hide relevant psychological disorders that will require specialized evaluation(14).
Drug-induced acne
It is not a true acne as it is a monomorphic process without comedones, which is why it is referred to as drug-induced acneiform eruptions. Its onset is chronologically related to the start of the treatment involved. Many drugs can be involved in its appearance, the most common being corticosteroids (Fig. 3), but also: antiepileptic drugs, antituberculosis drugs, lithium, vitamin B, halogenated compounds (iodides or bromides) and epidermal growth factor receptor inhibitors, among others.
Figure 3. Monomorphic drug acne (oral corticosteroids).
Endocrine acne
The term is used in those women who besides acne associate other manifestations of hyperandrogenism, and where polycystic ovary syndrome is the most frequent condition. SAHA syndrome (Seborrhea, Acne, Hirsutism and Alopecia) is an acronym for the main manifestations of this dermatological androgenization entity(15). Hormonal investigations are necessary for its diagnosis and management.
Acne induced by topical substances
It includes different clinical forms, notably cosmetic acne, which predominates in women and is related to the use of comedogenic cosmetics. Also, excessive washing as an attempt to improve acne can aggravate it (detergent acne).
Mechanical acne
It refers to the appearance of acne lesions in areas subjected to prolonged friction. Continuous irritation of the superficial area of the pilosebaceous duct and excessive hydration of the area due to occlusion, would be the factors involved in its appearance. The distribution of lesions and the mechanical history facilitates the diagnosis. A current example would be acne exacerbated or triggered by the use of protective face masks(16), which was interestingly already referred to in Asian literature in a previous coronavirus epidemic(17).
Hidradenitis suppurativa
It is an inflammatory process with a chronic and recurrent course, which predominantly affects: armpits, groin and anogenital area, sometimes associated with severe inflammatory acne with which, as mentioned previously, it shares physiopathogenic mechanisms(18). It usually begins during puberty, although there are childhood cases and its etiopathogenesis involves familial, endocrine factors (obesity, hyperandrogenism) and local irritation(18). It is characterized by: comedonal lesions, papules, pustules, and painful nodules that converge into large abscesses with sinus discharge and a tendency to cicatrize in the areas previously described.
Diagnosis
The diagnosis is based on clinical examination. The use of scales to determine the predominant type of lesion and its severity will guide the treatment.
The diagnosis of acne is a clinical one, based on the presence of comedones and/or inflammatory lesions. Examination with Wood’s light will allow to observe the presence of C. acnes in the comedones, as it emits a red-orange fluorescence due to the production of porphyrins (Fig. 4). Only in case of suspected endocrine acne, will complementary examinations be carried out to rule out hyperandrogenism.
Figure 4. Orange follicular fluorescence: relationship with porphyrins produced by C acnes. This indicates that the patient does not undergo treatment or that the microorganism is resistant.
In addition to the predominant type of lesion and its location, there are multiple systems for assessing the severity of acne(19,20), although they are of limited use in daily practice. The simplest is a qualitative classification, taking into account the type of dominant lesion(1) (Table I), which can be completed according to the severity (mild, moderate, severe).
Treatment
The treatment of choice will be combined topical products containing antibiotics, retinoids, or benzoyl peroxide; while, in case of non-response or moderate-severe acne, oral treatment with antibiotics or isotretinoin will be prescribed.
Basic concepts
The treatment of acne should begin by knowing the clinical history and response to other drugs that have been administered to the patient, in order to design an individualized plan for maximum effectiveness. Low adherence to treatment sometimes conditions results not turning out as expected, hence it is essential to indicate and explain some of the most relevant aspects of this, specifically, on the progress of the process, which may require treatment during months or years.
Compliance improvement is increased by using topical products with fixed combinations, rather than multiple products separately. Combination products have two advantages: on the one hand, they act against several pathogenic factors of acne simultaneously and, on the other, they simplify the therapeutic regimen. Patients should be informed that clinical improvement will not be achieved immediately, but rather after 6-8 weeks of treatment.
In order to minimize irritation from topical treatments, it should be indicated that the initial application can be gradual to improve tolerance during the first few weeks. We must instruct the patient in the use of non-comedogenic moisturizers, along with the avoidance of products that contribute to the development of acne. The use of a mild daily wash soap is advisable, while excessive hygiene can alter the skin barrier and increase the irritation potential of the treatment.
In the follow-up clinics, the therapeutic plan should be reinforced and inquire the presence of possible triggers or aggravating factors. In patients refractory to treatment, folliculitis due to gram-negative organisms should be excluded, especially in patients receiving continuous antibiotic treatment. Regarding diet, as mentioned above, it is not necessary to make dietary restrictions, except in case the patient relates acne appearance to certain foods or in case of deterioration with the consumption of abundant skimmed milk, or foods with a high sugar load.
The main objective of acne treatment is to avoid the appearance of scars, so the treatment must be as early as possible, with treatment modality appropriate to the severity and extension. Table II shows a therapeutic algorithm(1) to be followed in patients with acne. In general, the use of an oral or topical antibiotic in monotherapy should be avoided, in order to reduce the risk of bacterial resistance.
Topical treatment
Topical treatment will be prescribed in all patients with acne, in monotherapy in mild acne and, combined with systemic treatment, in cases of moderate or severe acne. In addition, after systemic treatment, maintenance topical retinoids should be applied. The patient should be insisted on applying it not only to the lesions, but also to the areas susceptible of presenting them, so as to avoid their appearance.
Retinoids
Topical retinoids prevent the formation of comedones and inflammatory lesions, normalize the desquamation of keratinocytes, and have an anti-inflammatory effect. They include: retinoic acid, isotretinoin, adapalene, tazarotene (in Spain this is indicated for the treatment of psoriasis) and, recently, tripharotene. The latter has been approved as a 0.005% cream, being a fourth-generation retinoid for the treatment of acne vulgaris on the face and trunk, from 12 years of age onwards. Tripharotene selectively targets gamma retinoic acid (RAR-γ) receptors, the most common ones in the skin(21).
Topical retinoids also enhance the penetration of associated topical antimicrobials, thereby increasing their efficacy. They are used as monotherapy for comedonal acne and as part of a combination therapy for mild-moderate papule-pustular acne(22). They achieve a 40-70% reduction in comedones and inflammatory lesions(23). They are also recommended as a maintenance treatment to prevent recurrences. Different concentrations are used, depending on severity and clinical tolerance, in aqueous creams or gels. The recommendation is to start with low concentrations or short application times, and gradually increase according to tolerance.
The most frequent side effects are: irritant dermatitis and photosensitivity. In 20% of patients, a transient increase in inflammatory lesions may occur. It is contraindicated in pregnancy due to its proven teratogenic properties with oral administration.
Benzoyl peroxide
It is an antimicrobial agent with anti-inflammatory and comedolytic activity, its main action being the neutralization of C. acnes in the hair follicles, thus achieving a bacteriostatic and possibly bactericidal effect, similar to topical antibiotics and without being associated with antimicrobial resistance. It is marketed in different concentrations (from 2.5% to 10%) and galenic forms (creams, gels, cleansers), alone or in combination with other active ingredients.
Its main side effects are irritation, in addition to discoloring dark clothes or hair. Tolerance increases if applied not immediately after the skin wash, but minutes later. It can be used during pregnancy and lactation.
Topical antibiotics
They have antibacterial action, inhibiting the growth and activity of C. acnes and a direct and indirect anti-inflammatory effect. They are used as part of combination treatments for mild-moderate papulopustular acne.
The most frequently used are clindamycin and erythromycin, in concentrations of 1-4%. Nadifloxacin 1% is a topical quinolone that also appears to have certain antiandrogenic action in vitro, in addition to inhibiting the activation of T cells and keratinocytes.
Monotherapy use is not recommended, due to the possible development of resistance to antibiotics and the slower onset of action. In the case of macrolides, resistances greater than 50% of strains of C. acnes have been reported in some countries(24). For this reason, they should be discontinued once improvement is appreciated and, in case of ineffectiveness after 6 to 8 weeks, when another treatment should be considered. The combination of topical antibiotic with topical retinoid or benzoyl peroxide reduces the possibility of resistance(22).
Side effects are less frequent compared to those of oral antibiotics and generally mild and local (pruritus, xerosis). Pseudomembranous colitis is a rare complication associated with topical clindamycin. Nadifloxacin should not be used in children under 14 years of age, as it is a fluoroquinolone.
Azelaic acid
Azelaic acid is a dicarboxylic acid that exhibits antimicrobial and anti-comedogenic activity(25). It is used for comedonal and inflammatory acne. It is available as 20% cream and 15% gel, and its main side effect is mild irritation. It can be used in pregnancy and lactation.
Other topical treatments
Salicylic acid is used as a comedolytic and antibacterial. It is used in various galenic presentations at 0.5-2%, in non-inflammatory forms, being less effective than topical retinoids. Its main side effects are: erythema and scaling.
The α-hydroxy acids are used in the comedonian forms at various concentrations and, in general, with good tolerance. The most used one is glycolic acid, which can be found in presentations combined with tretinoin and topical clindamycin.
Niacinamide is the active form of vitamin B3, with anti-inflammatory properties. It has been used as 4% hydroalcoholic gel in the treatment of mild acne.
Combined treatment
Combinations with retinoids are recommended as a first line of treatment for mild-moderate and papulopustular acne. Combinations of various therapeutic agents allow to target multiple pathogenic factors of acne. On the other hand, the combination of antibiotics with benzoyl peroxide reduces the possibility of bacterial resistance. Marketed fixed-dose combinations include the following:
• 0.1% adapalene plus 5% benzoyl peroxide, applied daily as a gel.
• Clindamycin 1% plus benzoyl peroxide 5% gel, which is more effective than both products used separately.
• 0.025% tretinoin plus 1% clindamycin gel.
• 0.02% tretinoin plus 4% glycolic acid, plus 0.8% clindamycin gel.
Systemic treatment
Indicated in patients with moderate-severe acne or in the absence of response to topical treatment in mild or mild-moderate acne. Also indicated in cases of extensive skin involvement.
Hormonal treatment
Most combined contraceptives (estrogen + progestogen) have the ability to improve acne and hirsutism to a greater or lesser extent, which is why they are useful in the patient with hyperandrogenism, but it is also an effective treatment in women with acne, regardless of androgen serum concentrations. Contraceptives improve: seborrhea, androgenic alopecia, SAHA syndrome and late-onset acne, in addition to regulating menstrual disturbances. Its main objective is to offset the effect of androgens on the sebaceous glands.
Its main indications in patients with acne are: failure of antibiotic treatment, when oral isotretinoin is contraindicated or inappropriate, and if, in addition to acne, it is necessary to control the menstrual cycle or as a contraceptive method. Better results seem to be obtained, even with normal serum androgen levels, in women with predominantly inflammatory lesions on the lower half of the face and neck who frequently present with premenstrual exacerbations of their acne.
Thrombosis is one of the most serious side effects of oral contraceptives which depends, over all, on the estrogens used and their dose. In order to reduce this side effect, the oral contraceptive of choice should be one that combines 30 µg or less of ethinyl estradiol (estrogen) with gestagen. If our aim is to treat signs of androgenization, the oral contraceptives of choice would be those with progestogens with an antiandrogenic effect: cyproterone acetate, chlormadinone acetate, drospirenone, dienogest, and nomegestrol acetate(26). According to a meta-analysis, cyproterone acetate and drospirenone were found to be the most effective progestogens in treating acne(27). The response is observed from the third month of treatment, and it should be maintained for a year, in the absence of side effects, with risk of recurrence upon discontinuation, thus its association to other treatments.
Oral antibiotics
They are used in moderate-severe inflammatory acne. They produce an anti-inflammatory action through the inhibition of the growth of C. acnes, in addition to reducing the amount of free fatty acids and, thus, their irritating effect. The most used ones are: oral tetracyclines (doxycycline, minocycline) and macrolides (erythromycin, azithromycin and josamycin). Doxycycline is the most widely used one, with doses ranging between 50-100 mg per day, the most frequent side effect being photosensitivity. They are generally well tolerated, and serious side effects are rare. Tetracyclines should not be used in children under 8 years of age, due to their effects on the developing skeleton and dentition.
Treatments are usually maintained for at least a month and a half, because as with other treatments, its clinical effect takes weeks to be appreciated, reducing the dose or suspending treatment once the appearance of inflammatory lesions diminishes, usually after 3 months.
They should be avoided as monotherapy, associating topical retinoids or benzoyl peroxide, if necessary: repeat treatment, use the same antibiotic if it was effective, and avoid the simultaneous use of topical and oral antibiotics, so as not to favor the appearance of resistance.
Oral isotretinoin
13-cis-retinoic acid of vitamin A or isotretinoin is indicated in severe nodule-cystic acne, refractory to other treatments or that can lead to scarring. It constitutes the therapeutic pillar of severe acne, but also for acne that has an important impact on the quality of life of the patient: inflammatory acne resistant to conventional treatment and chronic forms of recurrent tendency, in addition to gram-negative folliculitis, facial pyoderma and severe forms of rosacea.
It acts on all the factors involved in the pathophysiology of acne.
It is usually administered at doses of 0.5-0.6 mg/kg per day (from 0.1 to 2 mg/kg/day), maintaining the treatment until reaching a total dose of 120 to 150 mg/kg, so as to reduce the possibility of relapses. Doses can be modified based on clinical response and side effects. It should be taken with meals to enhance its absorption. Low-dose isotretinoin regimens (0.25-0.4 mg/kg/day) are effective, with fewer side effects, but higher recurrence rates(28).
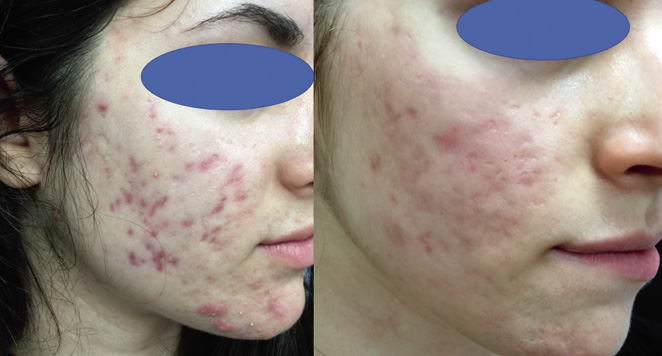
The clinical response is excellent (Fig. 5), most of the cases responding to a single 6-month course, although, in general, the results are not evident until 1-2 months after starting the treatment, a period in which even a certain exacerbation can be observed.
Figure 5. Deep inflammatory acne: before and after treatment with isotretinoin.
Side effects depend on the administered dose and, in general, are similar to those of hypervitaminosis A. Almost all patients present with cheilitis and, more than 50%, cutaneous and mucosal xerosis. Xerophthalmia, alteration of night vision, conjunctivitis, keratitis, headache and epistaxis can also be observed. Myalgias can affect 15% of patients, being the most frequent musculoskeletal manifestations. Rarely, long-term hyperostosis or osteoporosis has been described, hence radiological studies are not indicated in standard treatments. Exceptionally, an association with inflammatory bowel disease has been described, without finding, to date, an increased risk in patients treated with isotretinoin(29). The relationship with diabetes has been studied, identifying that treatment with isotretinoin significantly increases the level of serum adiponectin, but does not alter the state of insulin resistance in patients with acne(30). Its concomitant administration with tetracyclines is contraindicated, due to the increased risk of benign intracranial hypertension.
Digestive symptoms are rare. More frequent is the elevation of transaminases that can appear in 15% of the cases. In addition, triglycerides can increase in up to 25% of patients, especially in the first month, and they tend to decrease when the dose decreases. Triglyceride concentrations beyond 700-800 mg/dl is a criterion for stopping treatment. Full blood count can show: anemia, leukopenia, thrombocytosis or thrombopenia, and elevated ESR.
Psychiatric side effects such as: increased risk of depression, suicide, psychosis, and aggressive and violent behaviors have been described, although the causal relationship or mechanism of action remains unclear. A prevalence of depression of 1-11% has been described in patients receiving isotretinoin, a similar percentage to that observed in patients receiving oral antibiotic treatment, so it cannot be established as a cause(31). Despite this, the majority of patients experience improvement in the psychosocial repercussions related to acne, once treatment has begun. In any case, it is important that the patient and his family are aware of these data, in addition to identifying patients at risk and the possible appearance of any related symptoms. In case of severe headache, abnormal night vision or psychiatric manifestations, isotretinoin treatment must be stopped.
Isotretinoin is teratogenic, producing its maximum effect in the third week of gestation, so women of childbearing age should not start treatment until they have a negative pregnancy test, which should be repeated monthly. It is essential to recommend contraceptive methods from 1 month before to 1 month after the end of treatment. Patients must be adequately informed and a specific informed consent document should be completed.
The patient should be informed of the therapeutic effects of isotretinoin and taught to control or prevent side effects (adequate hydration, artificial tears to control dry eyes and nose, avoid irritants, elude alcoholic beverages, photoprotection, avert blood donation during the treatment…).
Recurrence of acne is not uncommon, in most patients with a good response to conventional therapy, but in some cases, a new cycle of treatment with isotretinoin must be indicated. It usually happens in the first year after treatment, and is rare after 3 years. Factors associated with risk of recurrence include: a low daily dose of isotretinoin (0.1-0.5 mg/kg) or not reaching a certain total dose (120-150 mg/kg), severe or prolonged acne, women of more than 25 years at the onset of treatment, endocrine abnormalities, patients under 16 years of age and acne located on the trunk. Maintenance treatment with topical retinoids can reduce recurrence risk by avoiding the formation of microcomedones.
The investigations to be carried out prior to prescribing the treatment include: full blood count, liver function tests, triglyceride concentrations and a urine pregnancy test in the case of females. The next check-up, including liver function and lipid profile, should take place after a month; if everything remains within normality, it is not necessary to repeat them, as long as we are dealing with a patient on usual doses and without other underlying pathology(32-33).
In conclusion, it is fair to say that isotretinoin is an effective medication, which can achieve the definitive “cure” in a high percentage of cases, with a comfortable administration and known dosage, and with controllable side effects which, in general, are well tolerated.
Other treatments
Phototherapy
Phototherapy targets C. acnes, a producer of porphyrins, especially coproporphyrin III. These porphyrins can be activated by light, at specific wavelengths (blue light at 415 nm and mixed blue-red light at 415 and 660 nm), producing a photodynamic effect capable of destroying these bacteria. It has been used in mild or moderate forms of inflammatory acne, with variable response.
Photodynamic therapy
The combination of topical aminolevulinic acid or methylaminolevulinate, metabolized to protoporphyrin IX in the pilosebaceous unit, a powerful photosensitizer, and the subsequent activation by a light source with a specific wavelength, would lead to: reduction of sebaceous secretion, destruction of C. acnes and also decreased ductal hyperkeratinization.
Intense pulsed light and laser
The pulsed dye laser at 585 nm, the neodymium: YAG at 1320 nm and the 1450 nm diode laser have been used in inflammatory acne, but with transient improvement, which together with the discomfort that it generates, makes it of limited performance.
Zinc
It does not improve comedones, but it does show some efficacy in treating inflammatory acne. Its action is due to: inhibition of neutrophil chemotaxis, inhibition of 5α-reductase and tumor necrosis factor. Oral route (200 mg/day, administered outside of meals) has been indicated. Its main side effects are gastrointestinal (nausea, vomiting, epigastric pain).
Corticosteroids
Its use is associated with the appearance of acne lesions (corticoid drug acne); however, they may be indicated in certain cases. Thus, in severe inflammatory forms, liable to exacerbation at the beginning of treatment with isotretinoin, a short course of oral corticosteroids rapidly reduces the number of lesions and the risk of exacerbating them with retinoid. Similarly, intralesional injection of corticosteroids may be useful in large, recent-onset inflammatory lesions.
Adjuvant treatment
Comedone removal
This technique achieves immediate improvement, which must be maintained with topical retinoids that prevent the formation of new microcomedones. Macrocomedones are a cause of therapeutic failure and do not usually respond to treatment with topical or oral retinoids; hence the need to eliminate them.
Chemical exfoliation (“chemical peels”)
It is indicated once acne is controlled, for the treatment of superficial scars or residual hyperpigmentation. The following are used: α-hydroxy acids (especially glycolic acid), trichloroacetic acid or salicylic acid.
Cryotherapy
Cryotherapy of liquid nitrogen and spray applicators has been used, especially in scar lesions.
Surgery
Surgery can be performed in case of scar lesions. It would include: removal of depressed scars (“lift technique”), classic dermabrasion, the use of filling material in deep scars with a non-fibrotic base or laser therapy.
Role of the Primary Care pediatrician
Acne in the majority of patients can be managed in the Primary Care setting. A correct treatment includes: early onset and appropriate therapy to the type of acne, use of combined treatments, never use antibiotics in monotherapy nor associate topical and oral antibiotics and, in the follow-up, reinforce the therapeutic plan and assess the response and tolerance to the treatment. Referral to the dermatologist would be considered in the following cases:
• Severe forms of acne.
• Moderate forms without response to prescribed topical and/or oral treatments.
• Patients with important psychosocial repercussions of the disease.
• Suspicion of an associated underlying endocrine disorder.
• Although rare, differential diagnosis with other processes.
• Finally, whenever treatment with isotretinoin is considered, as it is a drug that requires prescription by a specialist and a medical inspector visa. In these cases, the follow-up will be coordinated with the specialist.
Bibliography
The asterisks show the interest of the article in the opinion of the authors.
1.*** Zaenglein AL, Pathy AL, Schlosser BJ, Alikhan A, Baldwin HE, Berson DS, et al. Guidelines of care for the management of acne vulgaris. J Am Acad Dermatol. 2016; 74: 945-73.
2. Guerra A. Estudio epidemiológico descriptivo transversal sobre la prevalencia del acné en la población adolescente española. Act Dermatol. 2001; 11: 1-6.
3. Heng AHS, Chew FT. Systematic review of the epidemiology of acne vulgaris. Sci Rep. 2020; 10: 5754.
4. Lichtenberger R, Simpson MA, Smith C, Barker J, Navarini AA. Genetic architecture of acne vulgaris. J Eur Acad Dermatol Venereol. 2017; 31: 1978-90.
5. González-Cantero A, Arias-Santiago S, Buendía-Eisman A, Molina-Leyva A, Gilaberte Y, Fernández-Crehuet P, et al. ¿Existe variación en los diagnósticos dermatológicos entre la temporada de frío vs calor? Un subanálisis del estudio DIADERM (España 2016). Actas Dermosifiliogr. 2019; 110: 734-43.
6. Zaenglein AL. Acne vulgaris. N Engl J Med. 2018; 379: 1343-52.
7. Scholz CF, Kilian M. The natural history of cutaneous propionibacteria, and reclassification of selected species within the genus Propionibacterium to the proposed novel genera Acidipropionibacterium gen.nov., Cutibacterium gen.nov. and Propionibacterium gen.nov. Int J Syst Evol Microbiol. 2016; 66: 4422-32.
8.*** Dréno B, Pécastaings S, Corvec S, Veraldi S, Kharmari A, Roques C. Cutibacterium acnes (Propionibacterium acnes) and acne vulgaris: a brief look at the latest updates. J Eur Acad Dermato Venereol. 2018; 32: 5-14.
9. Pécastaings S, Roques C, Nocera Th, Peraud C, Mengeaud V, Khamari A, et al. Characterisation of Cutibacterium acnes phylotypes in acne and in vivo exploratory evaluation of Myrtacine. J Eur Acad Dermatol Venereol. 2018; 32: 15-23.
10. Claudel JP, Auffret N, Leccia MT, Poli F, Dréno B. Acne and nutrition: hypothesis, myths and facts. J Eur Acad Dermatol. 2018: 32: 1631-7.
11. Dréno B, Bettoli V, Araviiskaia E, Sánchez Viera M, Bouloc A. The influence of exposome on acne. J Eur Acad Dermatol. 2018; 32: 812-9.
12. Bocquet-Trémoureux S, Corvec S, Khammari A, Daqniele MA, Boisrobert A, Drenó B. Acne Fulminans and Cutibacterium acnes phylotypes. J Eur Acad Dermatol. 2020; 34: 827-33.
13. Miller IM Echeverria B, Torrelo A, Jemec GB. Infantile acne treated with oral isotretinoin. Pediatr Dermatol. 2013; 30: 513-18.
14. Anzengruber F, Ruhwinkel K, Ghosh A, Klaghofer R, Lang UE, Navarini AA. Wide range of age of onset and low referral rates to psychiatry in a large cohort of acne excoriée at a Swiss tertiary hospital. J Dermatol Treat. 2018; 29: 277-80.
15. Orfanos CE, Adler YD, Zouboulis CC. The SAHA syndrome. Horm Res. 2000; 54: 251-8.
16. Han C, Shi J, Chen Y, Zhang Z. Increased flare of acne caused by long-time mask wearing during COVID-19 pandemic among general population. Dermatol Ther. 2020; 29: e13704.
17. Tan KT, Greaves MW. N95 acne. Int J Dermatol. 2004; 43: 522-3.
18. Bandera A, de Lucas R. Aspectos epidemiológicos, clínicos y terapéuticos en situaciones especiales: hidradenitis supurativa infantil. Actas Dermosifiliogr. 2016; 107: 51-60.
19. Puig L, Guerra-Tapia A, Conejo-Mir J, Toribio J, Berasategui C, Zsolt I. Validation of the Spanish Acne Severity Scale (Escala de Gravedad del Acné Española – EGAE). Eur J Dermatol. 2013; 23: 233-40.
20. 0’Brien SC, Lewis JB, Cunlife WJ. The Leeds revised acne grading system. J Dermatol Treat. 1998; 9: 215-20.
21. Scott LJ. Trifarotene: First Approval. Drugs. 2019; 79: 1905-9
22. Thiboutot D, Gollnick H, Bettoli V, Dréno B, Kang S, Leyden JJ, et al. Global Alliance to Improve Outcomes in Acne. New insights into the management of acne: an update from the Global Alliance to Improve Outcomes in Acne group. J Am Acad Dermatol. 2009; 60: S1-50.
23. Haider A, Shaw JC. Treatment of acne vulgaris. JAMA. 2004; 292: 726-35.
24. Walsh TR, Efthimiou J, Dréno B. Systematic review of antibiotic resistance in acne: an increasing topical and oral threat. Lancet Infect Dis. 2016; 16: e23-33.
25. Williams HC, Dellavalle RP, Garner S. Acne vulgaris. Lancet. 2012; 379: 361-72.
26. Requena C, Llombart B. Oral Contraceptives in Dermatology. Actas Dermosifiliogr. 2020; 111: 351-6.
27. Arowojolu AO, Gallo MF, López LM, Grimes DA. Combined oral contraceptive pills for treatment of acne. Cochrane Database Syst Rev. 2012; 7: CD004425.
28. Lee JW, Yoo KH, Park KY, Han TY, Li K, Seo SJ, et al. Effectiveness of conventional, low-dose and intermittent oral isotretinoin in the treatment of acne: a randomized, controlled comparative study. Br J Dermatol. 2011; 164: 1369-75.
29. Lee SY, Jamal MM, Nguyen ET, Bechtold ML, Nguyen DL. Does exposure to isotretinoin increase the risk for the development of inflammatory bowel disease? A meta-analysis. Eur J Gastroenterol Hepatol. 2016; 28: 210-6.
30. Sai TY, Liu HW, Chao YC, Huang YC. Effects of isotretinoin on glucose metabolism in patients with acne: A systematic review and meta-analysis. J Dtsch Dermatol Ges. 2020; 18: 539-45.
31. Huang YC, Cheng YC. Isotretinoin treatment for acne and risk of depression: A systematic review and meta-analysis. J Am Acad Dermatol. 2017; 76: 1068-76.e9.
32. Lee YH, Scharnitz TP, Muscat J, Chen A, Gupta-Elera G, Kirby JS. Laboratory Monitoring during Isotretinoin Therapy for Acne: A Systematic Review and Meta-analysis. JAMA Dermatol. 2016; 152: 3544.
33. Barbieri JS, Shin DB, Wang S, Margolis DJ, Takeshita J. The clinical utility of laboratory monitoring during isotretinoin therapy for acne and changes to monitoring practices over time. J Am Acad Dermatol. 2020; 82: 72-9.
34. de Lucas Laguna R. Acné. Pediatr Integral. 2016; XX(4): 227-33.
Recommended bibliography
– Zaenglein AL, Pathy AL, Schlosser BJ, Alikhan A, Baldwin HE, Berson DS, et al. Guidelines of care for the management of acne vulgaris. J Am Acad Dermatol. 2016; 74: 945-73.
Interesting article describing the current recommendations in the management of acne. They highlight the importance of topical retinoid in all patients with mild-moderate acne, combined either with benzoyl peroxide or topical antibiotic, always avoiding the latter in monotherapy, in order to reduce the risk of bacterial resistance. For moderate-severe acne, oral antibiotic treatment should be added, or in case of lack of response to it, oral isotretinoin in monotherapy should be started.
– Dréno B, Pécastaings S, Corvec S, Veraldi S, Kharmari A, Roques C. Cutibacterium acnes (Propionibacterium acnes) and acne vulgaris: a brief look at the latest updates. J Eur Acad Dermato Venereol. 2018; 32: 5-14.
The bacteria Cutibacterium acnes (formerly known as Propionibacterium acnes) is part of a healthy skin, however, it can also act as an opportunistic pathogen in the appearance of acne vulgaris. The novelties identified in the etiopathogenesis of acne place C. acnes in a different position than previously considered. The proliferation of C. acnes would not act as a trigger, since acne patients do not harbor more C. acnes in their follicles than normal individuals. Instead, the loss of microbial diversity in the skin, coupled with the activation of innate immunity, could lead to this chronic inflammatory condition.
| Clinical case |
|
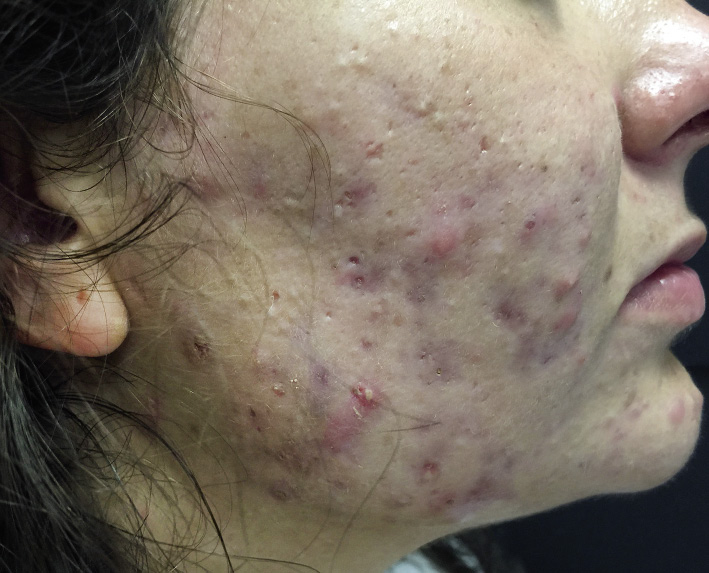
17-year-old female patient, with no relevant personal history. She presents facial injuries since a few years, which have experienced aggravation in recent months. She had been diagnosed of acne, and received treatment with doxycycline in monotherapy and various topical treatments later, with poor response. She had her menarche at age 11; and associates menstrual irregularities. On examination she presents: open and closed comedones, papules, pustules and nodules on the cheeks and, to a lesser extent on the forehead; seborrhea and hirsutism in the examined areas (scored 10 points on the Ferriman / Gallwey scale).
|
 Acne
Acne