|
| Topics on Continuous Training |
G. Domínguez-Ortega, N. Puente Ubierna
Gastroenterology and Nutrition Section. Pediatric Service. Niño Jesús University Children’s Hospital. Madrid
| Abstract
Eosinophilic gastrointestinal disorders (EGIDs) are a set of conditions with an increased number of eosinophils in any segment of the gastrointestinal tract without a known etiology. Clinical manifestations are based on the location and depth of the tissue eosinophilia. Eosinophilic Esophagitis is the most common one, although all EGIDs have an increasing prevalence. General Pediatricians should keep a high index of suspicion of these disorders and it is important to control symptoms and avoid complications. Diagnosis includes histologic criteria, so endoscopies and biopsies are always needed. Variable and individualized treatment is used. |
| Resumen
Los trastornos gastrointestinales eosinofílicos primarios son un conjunto de patologías crónicas que se caracterizan por un aumento del infiltrado eosinofílico, excluida una causa secundaria. Ocasionan síntomas variables según la localización del infiltrado. El más frecuente es la esofagitis eosinofílica, aunque la prevalencia de todos ellos está en aumento. Para el control de los síntomas y evitar sus posibles complicaciones es importante que el pediatra de Atención Primaria los conozca e incluya en el diagnóstico diferencial de patología digestiva. Dado que el diagnóstico incluye criterios histológicos, siempre va a ser necesaria la realización de una endoscopia con toma de biopsias. El tratamiento es variable e individualizado. |
Key words: Eosinophilic esophagitis; Eosinophilic gastritis; Eosinophilic enteritis; Eosinophilic colitis; Eosinophilic gastrointestinal disorders.
Palabras clave: Esofagitis eosinofílica; Gastritis eosinofílica; Enteritis eosinofílica; Colitis eosinofílica; Trastornos gastrointestinales eosinofílicos.
Pediatr Integral 2022; XXVII (2): 106 – 113
OBJECTIVES
• To update the understanding of the etiopathogenesis of primary eosinophilic gastrointestinal disorders.
• To identify the clinical symptoms of these disorders so as to make an early diagnosis.
• To learn the diagnostic criteria of these diseases and their differential diagnosis.
• To be aware of the different treatment options available in these pathologies.
Eosinophilic disease of the esophago-gastro-intestinal tract
Introduction
Eosinophilic gastrointestinal disorders (EGIDs) are characterized by eosinophilic infiltration in the esophagus, stomach, small intestine, or colon, in the absence of other causes of tissue eosinophilia.
The infiltration of eosinophils in the digestive mucosa is common, except in the esophagus, and its increase has been associated with different inflammatory pathologies of the digestive tract that cause dysfunction and digestive symptoms. These entities include: eosinophilic esophagitis (EoE), eosinophilic gastritis (EoG), eosinophilic enteritis (EoN), and eosinophilic colitis (EoC)(1).
In the first part of this review we will focus on EoE, the most prevalent entity of all EGIDs. In the second part we will focus on the other primary eosinophilic diseases of the digestive tract: EoG, EoN and EoC.
EGIDs are part of the group of gastrointestinal allergies (GIA). Other types of GIA have been addressed in other issues of this journal: No. 3 of 2020. Likewise, other types of esophagitis, beyond EoE, have been described in No. 8 of 2019 of this journal.
Eosinophilic esophagitis (EoE)
EoE is a chronic, immune-mediated disease characterized by a predominantly eosinophil inflammation exclusively at the esophageal level and by symptoms of esophageal dysfunction.
Epidemiology
It is an emerging disease and, currently, it is the most frequent cause of esophagitis after gastroesophageal reflux disease and the most frequent cause of dysphagia and impaction in children and young people.
The first cases were described in the late 1970s and it was defined as an entity in the early 1990s. Since then, numerous studies have been published on it, and its incidence and prevalence have increased exponentially(2). The prevalence of pediatric EoE in Spain in 2017 was 111.9 cases per 100,000 inhabitants and an incidence of 10.6 new cases per 100,000 inhabitants per year. It predominates in males, with a ratio of 3:1(3,4).
Patients with EoE have a high incidence of atopic diseases, such as: asthma, rhinoconjunctivitis, atopic dermatitis or food allergies.
Pathophysiology
EoE is a non-IgE-mediated gastrointestinal allergy. Predisposing genetic variants have been identified that, together with environmental interactions, can trigger the disease.
EoE is highly hereditary, with 64 times higher risk of developing it among siblings(5). The association between monozygotic twins is 58%, which implies a genetic determinant(6). Some studies have identified certain genes that contribute to the development of EoE (TSLP, CAPN14, EMSY, LRRC32, STAT6 and ANKRD27); however, it is important to highlight that dizygotic twins have a 36% association, while that of non-twin brothers is 2.4%. This significant difference would imply that environmental factors and epigenetic mechanisms influence its etiopathogenesis(6).
EoE is caused primarily, but not exclusively, by a non-IgE-mediated immune response to food antigens. In some patients, seasonal environmental allergy may play an important role(7). An abnormality of the epithelial barrier occurs with an increase in permeability, which facilitates the penetration of food antigens or pneumoallergens. Th2 lymphocytes, together with the proinflammatory cytokines, interleukin 5 (IL-5) and 13 (IL-13), induce eosinophilic infiltration in the esophageal mucosa(8).
A recent meta-analysis indicates that 3% of patients undergoing oral tolerance induction (OTI) to a specific food have esophageal biopsies compatible with EoE(9). These patients could have undiagnosed or subclinical disease, and it could have been exacerbated by immunotherapy.
In the same way as in other atopic diseases, digestive dysbiosis may play a role in the pathogenesis of EoE. Several studies have indicated differences between the esophageal microbiota of patients with EoE compared to healthy patients(10). More studies on the microbiota are necessary to understand the implications and its potential practical applications.
Clinical manifestations
The clinical manifestations are a consequence of the esophageal dysfunction and vary according to the age of the patient (Table I).
These patients develop compensatory behaviors for dysphagia, such as carefully chopping food before eating it, eating slowly, drinking water after each swallow, and avoiding foods with certain textures(11).
Diagnosis
The presence of compatible clinical symptoms (Table I) should raise suspicion of this entity. The confirmatory diagnosis is histological. Atopic comorbidity and family history support the suspicion.
The diagnostic criteria for EoE are:
• Presence of symptoms of esophageal dysfunction.
• Eosinophilic esophageal inflammation, with ≥ 15 eos/high-power field (HPF) (or ≥ 15 eosinophils/0.3 mm2 or ≥ 60 eosinophils/mm2).
• Exclusion of other causes of esophageal eosinophilia.
The British consensus guidelines on EoE, published in 2022, propose expressing eosinophilic infiltration as the number of eosinophils per 0.3 mm2, thus adapting to the current use of digital optical microscopy, which is replacing the analogue one(12).
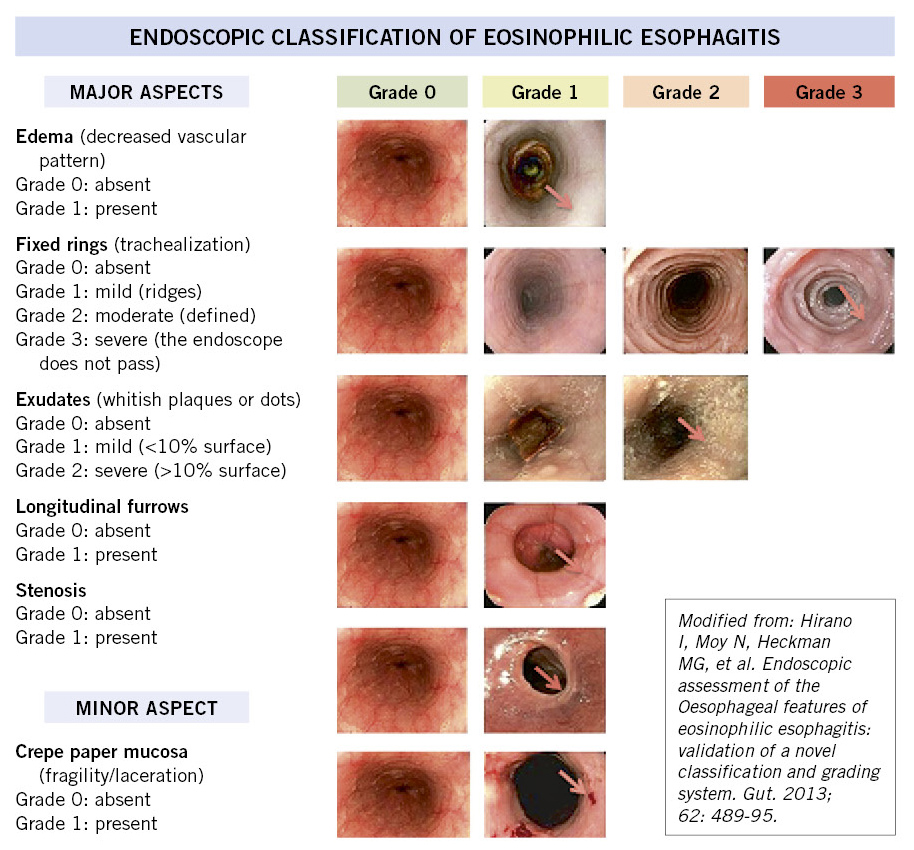
Diagnosis requires gastroscopy with biopsy. Gastroscopy allows visualizing the state of the esophageal mucosa and the presence of characteristics suggestive of EoE, such as: exudates, furrows, edema, strictures, rings, or the crêpe paper sign on the mucosa. The EoE Endoscopic Reference Score (EREFS) scoring system assesses these endoscopic characteristics (Fig. 1)(13).
Figure 1. Eosinophilic esophagitis scoring system: EoE Endoscopic Reference Score (EREFS).
Although the esophageal mucosa appears normal, biopsies must be performed. The involvement can be patchy, so it is recommended to take 6 biopsies from at least 2 locations in the esophagus, generally distal and mid-proximal. The consensus guidelines establish a threshold value of equal to or more than 15 eosinophils per high-power field (eos/HPF) in at least one of the esophageal biopsies to confirm the diagnosis(2).
In addition to the eosinophil peak count, there are other histological markers suggestive of this entity. The EoE Histology Scoring System has 8 histological characteristics that allow a more extensive evaluation of the esophageal mucosa(14): number of eosinophils, eosinophil abscesses, basal zone hyperplasia, dilated intercellular spaces, eosinophils in the superficial layer, dyskeratosis, abnormality of the superficial epithelium and fibrosis of the lamina propria.
Currently, there are no non-invasive biomarkers validated for the diagnosis and/or monitoring of EoE, although it is a field continuously developing(2).
Differential diagnosis
For the diagnosis of EoE, it is mandatory to rule out other pathologies that may present with tissue eosinophilia (Table II).
Treatment
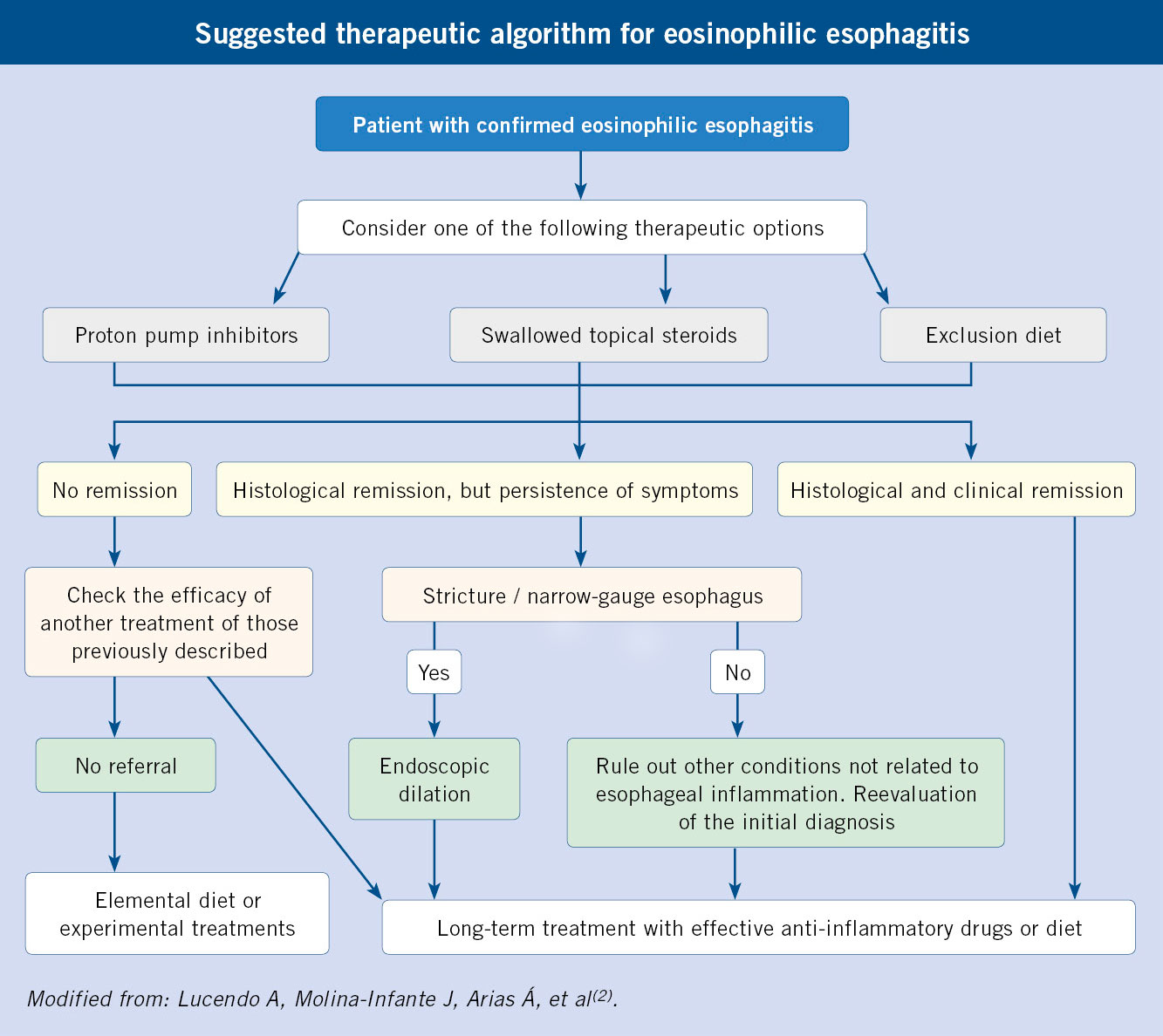
There are 3 treatment alternatives, all of them currently considered first-line: proton pump inhibitors (PPIs), swallowed topical corticosteroids (STCs), and empirical diet.
EoE is a chronic disease. Its natural history, without treatment, is associated with persistent symptoms. In addition, persistent esophageal inflammation causes esophageal remodeling that can lead to a fibrostenotic pattern. It is essential to establish early treatment to control symptoms, esophageal inflammation and prevent progression.
Current consensus guidelines establish three treatment alternatives: PPIs, swallowed corticosteroids, and an empirical diet, all of which are considered first choice(2). The option chosen must be individualized and agreed with the patient.
8-12 weeks after the start of treatment, a new gastroscopy should be performed with biopsies. This will allow evaluating the efficacy of the treatment and adjusting it in each patient. Clinical symptoms are not enough to assess the histological response; since, sometimes, there is not a good correlation between the symptoms and the histology.
Proton Pump Inhibitors (PPIs)
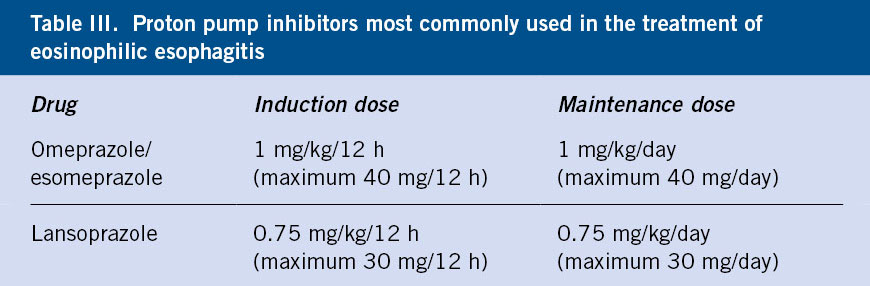
PPIs have an anti-inflammatory action at the esophageal level and a histological remission of 50% has been observed after treatment in children and adults(16). No differences have been seen between the different types of PPI (Table III). If they are effective, the dose will be gradually reduced until the minimum effective dose is found to maintain histological remission.
Swallowed topical corticosteroids
The histological remission rate is higher than the other treatment alternatives. Studies indicate remission rates of up to 85% at 12 weeks of treatment(17). Treatment with systemic corticosteroids should only be used in severe stenosis or dysphagia, due to increased side effects compared to topical treatments.
No drug specifically designed for EoE in children is yet on the market in Spain. Currently, the most commonly used corticosteroids are viscous oral budesonide in magistral compounded formula and liquid fluticasone, marketed for rhinitis (Flixonase® nasal solution) (Table IV).
Dietary treatment
Dietary treatment is the only one that addresses the cause of the disease. It eliminates the triggering food or foods from the diet so that long-term clinical and histological remission is achieved. It avoids the use of pharmacological treatments. Dietary treatment requires motivation on the patients´ part and their families, and in general there is worse adherence to the diet in adolescents.
• Elemental diet based on the exclusive administration of free amino acid formulas induces histological remission in 90% of patients. In many cases, administration through a nasogastric tube is necessary. Given the deterioration in the patient’s quality of life, it should only be recommended in severe cases and temporarily after lack of response to appropriate drug treatment or an empirical elimination diet. Targeted diets based on allergy testing are not recommended in EoE. The utility of food allergy tests to identify foods that trigger EoE is low(2).
• Empirical diet eliminates foods that have most often been linked to EoE, regardless of allergic testing. It always requires the performance of upper digestive endoscopies with biopsies after each dietary intervention, to evaluate the effect at the histological level. Initially, a 6-food elimination diet (cow’s milk, gluten, egg, soy, nuts, fish, and shellfish) was tested, with progressive sequential reintroduction. Given the difficulty in following these diets and the large number of endoscopies required, elimination diets of 4 foods (milk-gluten-egg-legumes) and then 2 foods (cow’s milk and gluten) were tested. More recently, a staggered approach has been proposed, starting with 2 foods, if there is no response, ascending to 4 and, finally, to 6. Currently, in Pediatrics, the empirical single food (milk) elimination diet is the recommended modality as the initial treatment, as it is much easier to implement. It achieves histological remission rates of up to 50% in pediatric patients, in addition to clinical and endoscopic improvement. In the absence of a response, the traditional pattern of empirical food elimination (2-4-6)(18) could be continued.
Endoscopic dilation
It is used in cases of severe esophageal stricture. In pediatric EoE, this procedure is exceptional, since in most cases it can be reversed with conventional treatment.
Dupilumab
Many biologic drugs have been studied; however, the only one approved by the FDA (United States Food and Drug Administration) in the US for EoE is Dupilumab. It is a monoclonal antibody directed against the interleukin 4 receptor α subunit (IL-4Rα) that blocks both IL-4 and IL-13 signaling and is not immunosuppressive. It is a drug previously approved for other pathologies such as atopic dermatitis and asthma, and since 2022 for pediatric EoE in children over 12 years of age weighing 40 kg or more. It is expected to be approved soon by the European Medicines Agency.
Other eosinophilic gastrointestinal disorders (EGIDs) besides EoE
EGIDs, beyond EoE, are also chronic immune-mediated disorders, clinically characterized by gastrointestinal (GI) symptoms and histologically by a pathologic increase in eosinophils in specific regions of the GI tract, in the absence of secondary causes of eosinophilia. They are named according to the location of the eosinophilic infiltrate. A recent consensus has established the proper nomenclature to refer to these disorders: eosinophilic gastritis (EoG), eosinophilic enteritis (EoN), and eosinophilic colitis (EoC). The term “eosinophilic gastroenteritis” (EGE), which generally involves the stomach and small intestine, has been widely used in the literature, although its use is currently recommended to be avoided(1).
Epidemiology
The prevalence of EGIDs other than EoE is low and predominates in females.
The prevalence of EoG in children is 4.4, EGE 10.7, and EoC 4.3/100,000. The prevalence of EoG increases with age, while the prevalence of EGE is higher in children under 5 years of age. The prevalence of EoC does not differ according to age(19).
In general, EGIDs other than EoE, are more frequent in women than in men, unlike EoE, with a prevalence of EoG in women of 7.9/100,000 vs. 5.4/100,000 cases in men(20). It can coexist with other atopic comorbidities and with EoE. The association with EoE is 10.6% in EoG, 12% in EGE and 10.9% in EoC(20).
Pathophysiology
The pathophysiology of EGIDs is only partially known. The combination of a genetic predisposition, the existence of dysbiosis and other environmental triggers (ingested or inhaled allergens) can lead to eosinophilic inflammation in patients with EGID.
Eosinophils induce bioactive inflammatory mediators that can trigger mast cell degranulation and release cytokines, lipid mediators, and neuromediators. These produce a Th2 immune response and intestinal inflammation. Recent studies seem to implicate Th2 cytokines (IL-4, IL-5 and IL-13) and eotaxin-3 in the pathogenesis of most of these diseases(21), although EoC seems to have a different transcriptome from the rest of EGIDs.
Clinical manifestations
EGIDs other than EoE, present a great diversity of symptoms that depend on the location in the gastrointestinal tract and the layer affected by tissue eosinophilia (Table V).
Diagnosis
Diagnostic criteria are based on a compatible clinical picture, an increased eosinophil count in biopsies, and the exclusion of secondary causes of gastrointestinal eosinophilia.
Diagnosis can be challenging, given the variability of the symptoms, the sometimes patchy involvement, the difficult access through endoscopic procedures on some occasions, or the possibility that the eosinophilic infiltrate only affects the deep layers, where biopsies cannot reach(23).
20-50% of patients with EGID have normal levels of eosinophils in peripheral blood, so a normal count is not enough to exclude the diagnosis. Recently, an endoscopic scoring system has been published to assess involvement in EoG with a strong correlation with histological results. The endoscopic findings evaluated are: erosions/ulcerations, nodularity, granularity, erythema, friability, fold thickening, and pyloric stenosis(24).
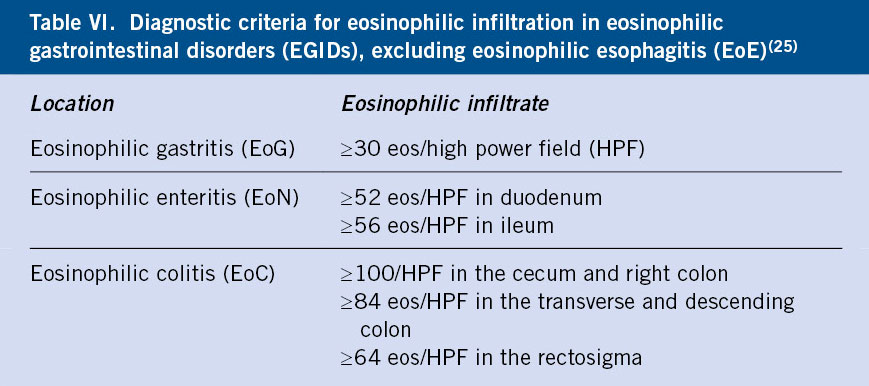
The presence of eosinophils is common in the digestive system except in the esophagus. The number of eosinophils in the rest of the sections of the gastrointestinal tract increases from proximal to distal, except in the colon, where it is higher in the ascending than in the descending(23). The diagnostic criteria for EGID include the finding of a higher than usual tissue eosinophilic infiltration for each region of the gastrointestinal tract according to recently established criteria (Table VI)(25).
A recent study established a high level of agreement between the histological and endoscopic findings of patients with EoG and a transcriptome-based panel of gastric biopsies and blood markers (eotoxin-3, IL-5, and thymic stromal lymphopoietin, among others). They do not replace the histological study of the samples, but the blood markers could serve as an initial non-invasive marker, and the study of the transcriptome can be used to confirm the diagnosis(26). At the moment, these markers are not available in routine clinical practice.
Differential diagnosis
Other causes of tissue eosinophilia must be ruled out (Table II), mainly inflammatory bowel disease (IBD).
In both EoC and IBD, eosinophilia can be found in the colonic lamina propria; however, IBD presents other acute and chronic histological changes that differentiate it from EoC. The clinical and endoscopic and histological findings must be considered to make the differential diagnosis.
Treatment
The low prevalence of EGIDs besides EoE, and their little-understood pathogenesis make it difficult to reach a consensus on its treatment.
No randomized controlled study on the treatment of EGIDs has been published, so there is not enough evidence available for the establishment of effective protocols.
More than a third of patients achieve spontaneous remission, but the majority will require dietary treatment and/or pharmacological intervention. Most patients treated with diets are pediatric, either using elemental or extensively hydrolyzed formulas or empirical diets eliminating a single food (milk or wheat) or combinations of foods considered to have a high risk of triggering an allergic response, as in the EoE, excluding also red meat in some studies(27).
The most commonly used pharmacological treatment is systemic glucocorticoids. Several corticosteroid treatment strategies have been described: oral prednisone and topical budesonide. Treatment with oral prednisone is started at an induction dose of 0.5-1 mg/kg/day until clinical remission is achieved. Subsequently, the dose is gradually decreased until it is discontinued or until a minimum maintenance dose that maintains clinical remission is achieved. Regarding treatment with topical budesonide, the induction dose is 9 mg/day with a subsequent decrease in dose(21). Rates of up to 42% of resistance or relapse have been published in a cohort of patients with EGE(28). Topical treatment is less effective than in EoE and systemic treatment has long-term side effects.
Among the biological drugs, cendakimab (anti-IL-13) and dupilumab (anti IL-4Rα) have been studied, showing that both decrease the eosinophil count and improve symptoms. Vedolizumab (binds to α4β7 integrin) has also been used successfully in some patients. Mepolizumab (anti-IL-5) and reslizumab (anti-IL-5) have shown a reduction in the number of eosinophils, but without clinical improvement. For EoG and EGE, both benralizumab (anti-IL-5) and lirentelimab or AK002 (anti-Siglec 8) have produced clinical and histological improvement(29).
EGID Prevention
There is not enough evidence to recommend any effective prevention measure for EGIDs.
Role of the primary care pediatrician
The role of the Primary Care pediatrician is essential in recognizing the symptoms of EGIDs, especially in atopic patients. EoE is the most common entity and should be suspected in a patient with compatible symptoms. Referral to the pediatric gastroenterologist should always be done, since a gastroscopy is necessary for diagnosis. In the case of EGIDs besides EoE, a diagnostic delay is common given the non-specific nature of the symptoms; however, in the event of alarm symptoms, referral to a pediatric gastroenterologist should be made to expedite the diagnostic and therapeutic process.
Conflict of interests
There is no conflict of interest in the preparation of the manuscript. Declaration of interests: none.
Bibliography
The asterisks reflect the interest of the article in the opinion of the authors.
1. Dellon E, Gonsalves N, Abonia JP, Alexander JA, Arva NC, Atkins D, et al. International Consensus Recommendations for Eosinophilic Gastrointestinal Disease Nomenclature Clinical Gastroenterology and Hepatology. 2022; 20: 2474-84.
2.*** Lucendo A, Molina-Infante J, Arias Á, von Arnim U, Bredenoord AJ, Bussmann Ch, et al. Guidelines on eosinophilic esophagitis: evidence-based statements and recommendations for diagnosis and management in children and adults. United European Gastroenterology Journal. 2017; 5: 335-58.
3. Arias A, Lucendo A. Incidence and prevalence of eosinophilic oesophagitis increase continuously in adults and children in Central Spain: A 12-year population-based study, Digestive and Liver Disease. 2019; 51: 55-62.
4. Barrio A, Botija Arcos G, Recio Linares A, García Rodríguez C. Esofagitis y gastritis. Pediatr Integral. 2019; XXIII: 377-85.
5. Collins MH. Histopathologic features of eosinophilic esophagitis. Gastroenterology Clinics of North America. 2014; 43: 25768.
6. Alexander ES, Martin LJ, Collins MH, Kottyan LC, Sucharew H, He H, et al. Twin and family studies reveal strong environmental and weaker genetic cues explaining heritability of eosinophilic esophagitis. J Allergy Clin Immunol. 2014; 134: 1084-92.
7. Jensen ET, Shah ND, Hoffman K, Sonnenberg A, Genta RM, Dellon ES. Seasonal variation in detection of oesophageal eosinophilia and eosinophilic oesophagitis. Aliment Pharmacol Ther. 2015; 42: 461-9.
8. O’Shea1 KM, Aceves SS, Dellon ES, Gupta SK, Spergel JM, Furuta GT, et al. Pathophysiology of Eosinophilic Esophagitis. Gastroenterology. 2018; 154: 333-45.
9. Lucendo AJ, Arias A, Tenias JM. Relation between eosinophilic esophagitis and oral immunotherapy for food allergy: a systematic review with meta-analysis. Ann Allergy Asthma Immunol. 2014; 113: 6249.
10. Mennini M, Tambucci R, Riccardi C, Rea F, De Angelis P, Fiocchi A, et al. Eosinophilic Esophagitis and Microbiota: State of the Art. Front. Immunol. 2021; 12: 595762.
11.*** Gutiérrez Junquera C, Fernández Fernández S, Domínguez-Ortega G, Vila Miravet V, García Puige R, García Romero R, et al. Recomendaciones para el diagnóstico y manejo práctico de la esofagitis eosinofílica pediátrica. An Pediatr (Barc). 2020; 92: 376.e1-376.e10.
12. Dhar A, Haboubi HN, Attwood SE, Auth MKH, Dunn JM, Sweis R, et al. British Society of Gastroenterology (BSG) and British Society of Paediatric Gastroenterology, Hepatology and Nutrition (BSPGHAN) joint consensus guidelines on the diagnosis and management of eosinophilic oesophagitis in children and adults. Gut. 2022; 71: 1459-87.
13. Hirano I, Moy N, Heckman MG, Thomas CS, Gonsalves N, Achem SR. Endoscopic assessment of the oesophageal features of eosinophilic oesophagitis: validation of a novel classification and grading system. Gut. 2013; 62: 489-95.
14. Collins MH, Martin LJ, Alexander ES, Boyd JT, Sheridan R, He H, et al. Newly developed and validated eosinophilic esophagitis histology scoring system and evidence that it outperforms peak eosinophil count for disease diagnosis and monitoring. Diseases of the Esophagus. 2017; 30: 1-8.
15. Naramore S, Gupta SK. Nonesophageal eosinophilic gastrointestinal disorders: clinical care and future directions. J Pediatr Gastroenterol Nutr. 2018; 67: 318-21.
16. Lucendo AJ, Arias Á, Molina-Infante J. Efficacy of proton pump inhibitor drugs for inducing clinical and histologic remission in patients with symptomatic esophageal eosinophilia: A systematic review and meta-analysis. Clin Gastroenterol Hepatol. 2016; 14: 13-22.
17. Lucendo AJ, Miehlke S, Schlag Ch, Vieth M, von Arnim U, Molina-Infante J, et al. Efficacy of Budesonide Orodispersible Tablets as Induction Therapy for Eosinophilic Esophagitis in a Randomized Placebo-Controlled Trial. Gastroenterology. 2019; 157: 74-86.e15.
18. Wechsler JB, Schwartz S, Arva NC, Kim KYA, Chen L, Makhija M, et al. A Single Food Milk Elimination Diet Is Effective for Treatment of Eosinophilic Esophagitis in Children. Clin Gastroenterol Hepatol. 2022; 20: 1748-56.e11.
19. Jensen ET, Martin CF, Kappelman MD, Dellon ES. Prevalence of Eosinophilic Gastritis, Gastroenteritis, and Colitis: Estimates From a National Administrative Database. J Pediatr Gastroenterol Nutr. 2016; 62: 36-42.
20.*** Naramore S, Gupta SK. Non-esophageal eosinophilic gastrointestinal disorders: clinical care and future directions. J Pediatr Gastroenterol Nutr. 2018; 67: 318-21.
21.*** Licari A, Votto M, D’Auria E, Castagnoli R, Caimmi SME, Marseglia GL. Eosinophilic Gastrointestinal Diseases in Children: A Practical Review. Current Pediatric Reviews. 2020; 16: 106-14.
22. Collins MH. Histopathology associated with eosinophilic gastrointestinal diseases. Immunol Allergy Clin North Am. 2009; 29: 109-17, x-xi.
23. Espín Jaime B. Patología digestiva eosinofílica. En: AEPap (ed.). Congreso de Actualización Pediatría 2019. Madrid: Lúa Ediciones. 3.0; 2019. p. 165-76.
24. Hirano I, Collins MH, King E, Sun Q, Chehade M, Abonia JP, et al. Prospective Endoscopic Activity Assessment for Eosinophilic Gastritis in a Multisite Cohort. Am J Gastroenterol. 2022; 117: 413-23.
25. Collins MH. Histopathologic features of eosinophilic esophagitis and eosinophilic gastrointestinal diseases. Gastroenterol Clin North Am. 2014; 43: 257-68.
26. Shoda T, Wen T, Caldwell JM, Collins MH, Besse JA, Osswald GA, et al. Molecular, endoscopic, histologic, and circulating biomarker-based diagnosis of eosinophilic gastritis: Multi-site study. J Allergy Clin Immunol. 2020; 145: 255-69.
27. Lucendo AJ, Serrano-Montalbán B, Arias Á, Olga Redondo O, Tenias JM. Efficacy of Dietary Treatment for Inducing Disease Remission in Eosinophilic Gastroenteritis. J Pediatr Gastroenterol Nutr. 2015; 61: 56-64.
28. Choi JS, Choi SJ, Lee KJ, Kim A, Yoo JK, Yang HR, et al. Clinical Manifestations and Treatment Outcomes of Eosinophilic Gastroenteritis in Children. Pediatr Gastroenterol Hepatol Nutr. 2015; 18: 253-60.
29. Dellon E, Spergel J. Biologics in eosinophilic gastrointestinal diseases. Ann Allergy Asthma Immunol. 2023; 130: 21-7.
Recommended bibliography
– Lucendo A, Molina-Infante J, Arias A, vonArnim U, Bredenoord AJ, Bussmann Ch, et al. Guidelines on eosinophilic esophagitis: evidence-based statements and recommendations for diagnosis and management in children and adults. United European Gastroenterology Journal. 2017; 5:335-58.
Consensus article between the main European Digestive and Allergology societies and working groups on the management of EoE.
– Gutiérrez Junquera C, Fernández Fernández S, Domínguez-Ortega G, Vila Miravet V, García Puige R, García Romero R, et al. Recomendaciones para el diagnóstico y manejo práctico de la esofagitis eosinofílica pediátrica. An Pediatr (Barc). 2020; 92: 376.e1-376.e10.
Updated guide for the management of EoE in Spain, prepared by the SEGHNP Working Group on Eosinophilic Gastrointestinal Disorders.
– Naramore S, Gupta SK. Non-esophageal eosinophilic gastrointestinal disorders: clinical care and future directions. J Pediatr Gastroenterol Nutr. 2018; 67:318-21.
Review article on EGIDs excluding EoE, which addresses everything from pathophysiology to clinical symptoms and diagnostic and therapeutic management. It promotes the creation and promotion of platforms for patients and professionals, such as CEGIR in the US.
– Licari A, Votto M, D’Auria E, Castagnoli R, Caimmi SME, Marseglia GL. Eosinophilic Gastrointestinal Diseases in Children: A Practical Review. Current Pediatric Reviews. 2020; 16: 106-14.
Updated article that summarizes the knowledge and management of EGIDs in Pediatrics. It describes the clinical features and diagnostic criteria, and addresses the differential diagnosis.
 Eosinophilic disease of the esophago-gastro-intestinal tract
Eosinophilic disease of the esophago-gastro-intestinal tract